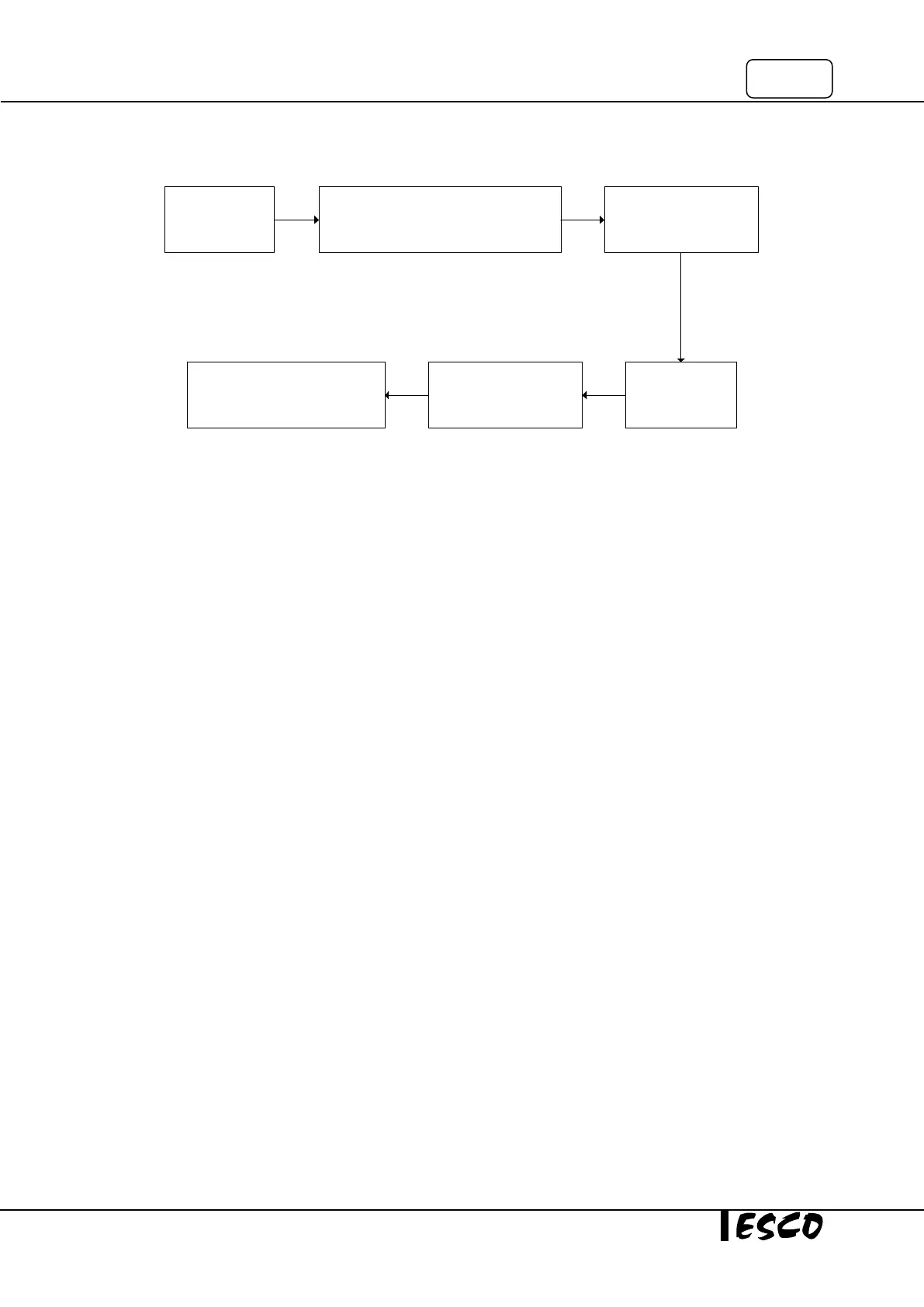
3.6 Completion Steps
Check the ammonia
concentration in
room air
Check the formaldehyde concentration in
room air at the end of decontamination cycle,
prior resuming work on the BSC
Check the formaldehyde and
ammonia concentration in
the BSC
Remove all
apparatus from
inside the BSC
Remove the
decontamination bag that
enclosed the BSC
Remove the disposible gown and
gloves carefully and wash hands using
germicidal soap thoroughly
During the procedure, until the area is determined to be safe, a full face gas
mask equipped with a formaldehyde canister should be worn. The filter for
the mask must be selected for the appropriate level of formaldehyde
exposure. It must also be replaced regularly following the manufacturer’s
instruction
3.7 Decontamination Validation Procedure (optional)
• Place some Bacillus subtilis var.niger spore strips inside the Unit prior to decontamination.
o Work surface/table tray
o Drain pan
o Downstream HEPA filter
• Place similar strips outside the decontamination room for positive control (optional).
• Remove all the spore strips and place in trypticase-soy broth and incubate for 2 days at 37°C.
• If there is no growth of bacteria inside the broth (the broth remains clear), the decontamination
procedure has been successfully verified.
 Loading...
Loading...