1
3
4B
2
4A
BEFORE USING THE DEVICE, PLEASE READ THE FOLLOWING
INSTRUCTIONS COMPLETELY AND CAREFULLY. CORRECT
APPLICATION IS VITAL TO THE PROPER FUNCTIONING OF THE
DEVICE.
5
1
2
3
4
5
7
2
31
ENGLISH
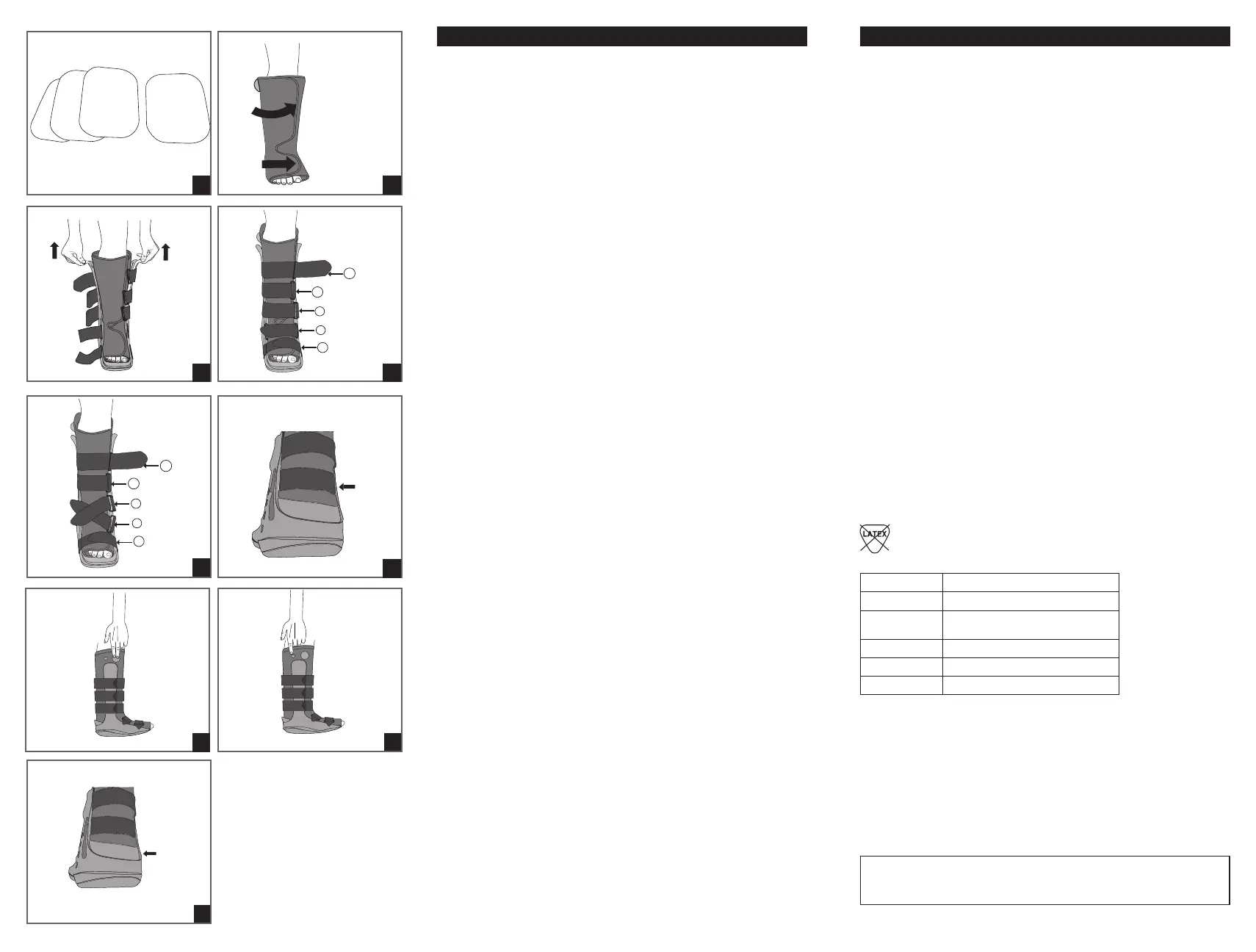
4. つま先から始めて脚の上方へ、すべてのブーツストラップを固定します。(
図4A)。
注 記:すべてのストラップは取り外し、交換が可能です。足首関節の2つ の ストラップ
は、患者のそれぞれのニーズに合わせて変更することができます。
足首交差ストラップ:交差ストラップを希望する場合は、ストラップ3を反対側の前
肢Dリ ン グ に 通 し ま す 。ス ト ラ ッ プ 2をストラップ 3上に交差し、反対側支柱のDリング
に通して固定します(
図4B)
。
5. ア キ レ ス 腱( 踵 ス ト ラ ッ プ )は 、ぴ っ た り と し て い る べ き で す が 、き つ す ぎ て は い け ま
せん。取り外すこともできます。(
図 5)
。
エアライナーの場合:
6. エアライナーを膨らませるには、ライナーが快適なクッション性とサポートを提供
するようになるまで、エアバルブを押して放す動作を数回繰り返します。過度に膨張
さ せ な い で く だ さ い(
図6)
。
7. ウォーカーライナーから空気を抜くには、収縮ボタンを押したまま、圧力が快適に
なるまで空気を抜きます。ライナーがきつすぎると感じる時は、空気を抜いてくださ
い。注意:製品の装着中にライナーがきつすぎると感じる場合は、収縮ボタンを押し
てライナーの空気を抜きます。直ちに担当の医療専門家に相談してください(
図7)
。
8. ウォーカーを取り外すには、すべてのストラップをゆるめ、ライナーの接触クロージ
ャを外します。
9. ウォーカーを再装着するには、ステップ2と 4に従います。
10. かかと部分がより広いウォーカーを希望する場合は、ギプス用のハサミを使ってソフト
インソールのかかと部分を切り取ることができます(
図8)
。
注記:再装着の際に適切にフィットさせるには、ブレースを取り外す前にエアセルの
空 気 を 抜 い てくださ い 。
注記:圧が高いほど歩行時のサポートは強く、座ったり横になったりする場合は低め
の圧力がより快適です。圧力は、ストラップを締めたり緩めたりして調整できます。
清浄手順:ライナーは、中性洗剤を用いて冷水中で手洗いします。自然乾燥させます。
注 記:すすぎが不十分だと、洗剤残留物による皮膚の炎症や製品材料が劣化する原因
になることがあります。
保管手順:本製品を使用するまでは、常温で保管してください。
保 証:DJO, LLCは、材料や工程の不具合に対して、購入日から 6 ヶ 月 間 に 限 り 、ユ ニ ッ
トおよびアクセサリのすべてまたは一部を修理あるいは交換します。本保証が現地規
制と矛盾する範囲については、当該現地規制の規定が適用されます。
特許出願中。
要処方。
1人の患者にのみ使用してください。
天然ゴムラテックス不使用。
材料成分:
コンポーネント 材料
フレーム 射出成形ポリプロピレン、アルミニウム
ライナー
フ ォ ー ム ラ ミ ネ ー ト 、ポ リ エ ス テ ル 、コ ッ ト
ン、ABS、PVC、シ リ コ ン 、ス チ ー ル 、TPU
外底 EVAフォーム/ラバー(ラテックスを含有しない)
中敷 EVAフォーム/ラバー(ラテックスを含有しない)
フック/ ループ ナイロン
INTENDED USER PROFILE: The intended user should be a licensed medical
professional, the patient or the patient’s caregiver. The user should be able to read,
understand and be physically capable of performing the directions, warnings and
cautions in the information for use.
INTENDED USE/INDICATIONS: The DonJoy Maxtrax 2.0 Air Walker is designed to
provide comfort, support and immobilization to the lower leg, ankle and foot. It may
be suitable for the treatment of soft tissue injuries of the lower leg such as acute
ankle sprains, stress fractures of the lower leg, stable fractures of the foot and/or
ankle including metatarsal fractures, Achilles tendon repairs and post-operatively
following bunion (Halux Valgus) surgery (modified Lapidus Fusion).
PERFORMANCE CHARACTERISTICS:
• Customizable fit designed to provide comfort to the limb or body segment.
• Soft-good/ semi-rigid designed to restrict motion through elastic or semi-rigid
construction.
• Providing immobilization or controlled movement to the limb or body segment.
CONTRAINDICATIONS: This device is contraindicated for unstable fractures of
the lower leg, ankle and foot. Do not use if you are allergic to any of the materials
contained within this product.
NOTE: Due to the molded outer sole, the orthopedic walker may be slightly higher
than a normal street shoe, therefore an athletic shoe or a street shoe with a ½”
heel, may be worn if necessary for patient comfort. A ProCare ShoeLift
TM
also may
be used on the shoe worn on the non-affected foot.
For Air liner, inflate and deflate the bladder once before use to ensure the integrity
of the air chamber.
WARNINGS AND CAUTIONS:
• This device is to be used under the supervision of a healthcare professional. The
determination of when to apply the brace and the frequency and duration of use
should be strictly at the discretion of the treating physician. Always consult with
your physician before making changes to the brace.
• Application of this device is recommended only when the fracture is
demonstrably stable and there are acceptable limits of angular and rotational
deformity.
• Do not use this device on patient’s incapable of communicating physical
discomfort.
• Check your skin frequently for “hot spots” and skin irritation.
• Do not use this device if you cannot feel sensations whether due to post-op
anaesthesia, nerve, skin or other conditions.
• Use caution when walking on slippery or wet surfaces to avoid injury.
• Do not use over open wounds.
• Do not over tighten straps. This may result in reduction of blood flow or
sensation.
• Do not over-inflate aircells. Improper aircell inflation may cause significant skin
irritation, especially in people with diabetic neuropathy. Reduce aircell pressure
with any visual skin changes or reported discomfort.
• Do not modify the device or use the device other than intended.
• If you develop an allergic reaction and/or experience itchy, red skin after coming
into contact with any part of this device, please stop using it and contact your
healthcare professional immediately.
• Do not use this device if it is damaged and/or the packaging has been opened.
• If pain, swelling, changes in sensation or other unusual reactions occur while
using this product, consult your medical professional immediately.
NOTE: Contact manufacturer and competent authority in case of a serious incident
arising due to usage of this device.
APPLICATION INFORMATION:
For first time application, loosen all straps and remove liner from boot.
1. Removable foam ankle pads are provided for patient comfort (
Figure 1
).
2. Place foot in liner and secure with contact closure. Make sure the heel fits
snugly into the posterior portion of the liner. Secure the forefoot flap on the liner
first followed by the lower leg portion. Make sure liner fits snug top to bottom
(
Figure 2)
.
お知らせ:最 大限の機能、強度、耐久性と快適性を得られるよう最先端技術によりあらゆる努力が払われていま
すが、本製品の使用によって怪我が防止されるという保証はありません。
6
8

 Loading...
Loading...