Biocompatibility and electrical
isolation
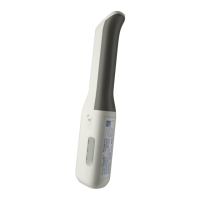
The autoinjector enclosure is
intended to come into contact with
the skin (see Guide to Parts). It is
a Type BF applied part. This
means that it is electrically
isolated from the battery. The
injection end and finger grip of
AutoTouch Connect
TM
are made of
ABS plastic. This material has
been biocompatibility-tested for
skin sensitivity and irritation.
Contraindications
This device is contraindicated for
use in a Magnetic Resonance (MR)
environment. For
contraindications of ENBREL
®
please refer to ENBREL prescribing
information.
 Loading...
Loading...