EN
Intended use
Medical Purpose
This product is intended to be used for inhaling medication for respiratory
disorders.
Intended User
• Legally certified medical experts, such as doctor, nurse and therapist.
• Caregiver or patient under the guidance of qualified medical experts for
home treatment.
• The user should also be capable of understanding general operation of
NE-C900 and the content of instruction manual.
Intended Patients
This product should not be used by patients, who are unconscious or are
not breathing spontaneously.
Environment
This product is intended for use in a medical facility, such as Hospital,
clinic and doctor’s office, and in a room of general household.
Durable period
Durable periods are as follows, provided the product is used to nebulize
2 ml of medication 10 times a day for 6 minutes each time at room
temperature (23°C).
Durable period may vary depending on usage environment.
Compressor (Main unit) 5 years
Nebulizer Kit 1 year
Mouthpiece 1 year
Nosepiece 1 year
Air Tube (PVC, 200cm) 1 year
Air Filter 60 days
Mask (PVC)/(SEBS) 1 year
Precautions for use
Warnings and cautions described in the instruction manual should be
observed.
Know your unit
Important safety instructions
Read all the information in the instruction manual and any other literature included in
the box before using the device.
Warning
:
Indicates a potentially hazardous situation which, if not avoided, could result in
serious injury.
(Usage)
• For type, dose, and regime of medication follow the instructions of your doctor or
respiratory therapist.
• If you feel anything unusual during use, stop using the device immediately and
consult your doctor.
• Do not use only water in the nebulizer for inhaling purposes.
• Keep the device out of the reach of unsupervised infants and children. The device
may contain small pieces that can be swallowed.
• Do not store the air tube while there is moisture or medication remaining inside.
• Do not use or store the device where it may be exposed to noxious fumes or volatile
substances.
• Do not use the device where it may be exposed to flammable gas.
• Do not cover the compressor with a blanket, towel, or any other type of cover during
use.
• Always dispose of any remaining medication after use, and use fresh medication each
time.
• Do not use in anaesthetic or ventilator breathing circuits.
• Do not block the ventilation slots. Never place the device where the ventilation slots
may be obstructed during operation.
• Store the parts in a clean location for avoiding infection.
(Risk of electrical shock)
• Never plug or unplug the device with wet hands.
• The compressor and power cord are not waterproof. Do not spill water or other liquids
on these parts. If liquid does spill on these parts, immediately unplug the
power cord
and wipe off the liquid with gauze or other absorbent material.
• Do not immerse the compressor in water or other liquid.
• Do not use or store the device in humid locations.
• Do not operate the device with a damaged power cord or plug.
• Keep the power cord away from heated surfaces.
(Cleaning and disinfecting)
Observe the rules below when cleaning or disinfecting parts. Failure to observe these
rules may result in damage, inefficient nebulization or infection. For the instructions,
refer to “Cleaning and disinfecting” section.
• Clean and disinfect the nebulizer kit, mask, mouthpiece or nosepiece before using
them for the first time after purchase.
• If the device has not been used for a long period of time, or if more than one person
uses the same device, clean and disinfect the nebulizer kit, mask, mouthpiece or
nosepiece before using them.
• Be sure to wash or wipe the parts after use, and ensure that they are thoroughly
disinfected and dried, and stored in a clean location.
• Do not leave the cleaning solution in the parts.
Caution:
Indicates a potentially hazardous situation which if not avoided, may result in
minor or moderate injury, or physical damage.
(Usage)
• Provide close supervision when this device is used by, on, or near children or invalids.
• Make sure that the parts are attached correctly.
• Make sure that the air filter is correctly attached.
• Make sure that the air filter is clean. If the air filter has been used for more than 60
days, replace it with a new one.
• Do not tilt the nebulizer kit at an angle of greater than 45 degrees in all directions or
shake it while in use.
• Do not use or store the device while the air tube is creased.
• Use only original nebulizing parts, air tube, air filter and air filter cover.
• Do not add more than 7 ml of medication to the medication tank.
• Do not carry or leave the nebulizer kit while the medication tank contains medication.
• Do not leave the device unattended with infants or persons who cannot express their
consent.
• Do not subject the device or the parts to any strong shocks such as dropping the device
on the floor.
• Do not jab the nozzle of the medication tank with a pin or any sharp object.
• Do not insert fingers or objects inside the compressor.
• Do not disassemble or attempt to repair the compressor or power cord.
• Do not leave the device or its parts in extreme hot or cold temperature, or under direct
sunlight.
• Do not block the air filter cover.
• Do not use the device while sleeping or if drowsy.
• Approved for human use only.
• When using the device, the main unit may become hot.
• Do not touch the main unit for other than necessary operation such as turning off the
power while nebulizing.
• To avoid the medication residue on the face, be sure to wipe the face after removing
the mask.
• To avoid injury to the nose mucosa, do not squeeze the nosepiece into the back of
the nose.
• Do not use a damaged nebulizer kit, mouthpiece or nosepiece.
(Risk of electrical shock)
• Always unplug the device from the power outlet after use and before cleaning.
• Plug the device into the appropriate voltage outlet. Do not overload power outlets or
use extension cords.
• Do not misuse the power cord.
• Do not wind the power cord around the compressor.
• Changes or modifications not approved by OMRON HEALTHCARE will void the
user warranty.
• Do not pull the power cord strongly.
(Cleaning and disinfecting)
Observe the rules below when cleaning or disinfecting parts. Failure to observe these
rules may result in damage, inefficient nebulization or infection. For the instructions,
refer to “Cleaning and disinfecting” section.
• Do not use a microwave oven, dish dryer or hair dryer to dry the device or the parts.
• Do not use an autoclave, EOG gas sterilizer or low temperature plasma sterilizer.
• When disinfecting parts by boiling, make sure that the container does not boil dry.
Otherwise it may also result in fire.
Accessories
Name Model Order No.
Nebulizer Kit & Mouthpiece Set
NEB-NSET3-90E 9520555-5
Adult Mask (PVC)
C28-8-E 9956275-1
Child Mask (PVC)
C28-9-E 9956276-0
Infant Mask (PVC)
NEB-MSSP-E 9520250-5
Adult Mask (SEBS)
U10-1P-E 9956312-0
Child Mask (SEBS) Set
U10-2P-E 9956281-6
Mouthpiece
C28-3P-E 9956273-5
Nosepiece
C28-7-E 9956274-3
Air Tube (PVC, 200cm)
NEB-TP-L20E 9520606-3
Air Filters x 5
C28-2-E 9956271-9
Thank you for purchasing the OMRON COMP AIR Pro.
This product was developed in conjunction with respiratory therapists
for the successful treatment of asthma, chronic bronchitis, allergies
and other respiratory disorders.
This is a medical device. Operate the device only as instructed by
your doctor and/or respiratory therapist.
How the nebulizer kit works
The medication that is pumped up
through the medication channel is
mixed with compressed air which is
generated by a compressor pump.
The compressed air mixed with
medication is turned into fine particles
and sprayed when in contact with the
baffle.
Manufacturer OMRON HEALTHCARE Co., Ltd.
53, Kunotsubo, Terado-cho, Muko, Kyoto, 617-0002 JAPAN
EU-representative
OMRON HEALTHCARE EUROPE B.V.
Scorpius 33, 2132 LR Hoofddorp, THE NETHERLANDS
www.omron-healthcare.com
Production facility
OMRON (DALIAN) CO., LTD.
Dalian, CHINA
Subsidiary
OMRON HEALTHCARE UK LTD.
Opal Drive, Fox Milne, Milton Keynes, MK15 0DG, U.K.
OMRON MEDIZINTECHNIK HANDELSGESELLSCHAFT mbH
John-Deere-Str. 81a, 68163 Mannheim, GERMANY
www.omron-medizintechnik.de
OMRON SANTÉ FRANCE SAS
14, rue de Lisbonne, 93561 Rosny-sous-Bois Cedex, FRANCE
Made in China
General Safety Precautions:
• Inspect the device and parts before using them each time, and check that there are
no problems. In particular, be sure to check the following:
-That the nozzle or air tube are not damaged.
-That the nozzle is not blocked.
-That the compressor operates normally.
• When using this device, there will be some noise and vibration caused by the pump in
the compressor. There will also be some noise caused by the emission of compressed
air from the nebulizer kit. This is normal and does not indicate a malfunction.
• Operate the device only as intended. Do not use the device for any other purpose.
• Do not use the device at temperatures greater than +40°C.
• Make sure that the air tube is securely attached to the compressor and nebulizer kit,
and does not come loose. Twist the air tube plug slightly when inserting it into the air
tube connectors to avoid the air tube disconnecting during use.
• To completely isolate the device from the power source unplug the plug from the
power source.
Keep these instructions for future reference.
Important information regarding Electro Magnetic Compatibility (EMC)
With the increased number of electronic devices such as PC’s and mobile (cellular)
telephones, medical devices in use may be susceptible to electromagnetic interference
from other devices. Electromagnetic interference may result in incorrect operation of the
medical device and create a potentially unsafe situation.
Medical devices should also not interfere with other devices.
In order to regulate the requirements for EMC (Electro Magnetic Compatibility) with
the aim to prevent unsafe product situations, the EN60601-1-2:2007 standard has
been implemented. This standard defines the levels of immunity to electromagnetic
interferences as well as maximum levels of electromagnetic emissions for medical
devices.
This medical device manufactured by OMRON HEALTHCARE conforms to this
EN60601-1-2:2007 standard for both immunity and emissions.
Nevertheless, special precautions need to be observed:
• Do not use mobile (cellular) telephones and other devices, which generate strong
electrical or electromagnetic fields, near the medical device. This may result in
incorrect operation of the unit and create a potentially unsafe situation.
Recommendation is to keep a minimum distance of 7 m. Verify correct operation of
the device in case the distance is shorter.
Further documentation in accordance with EN60601-1-2:2007 is available at OMRON
HEALTHCARE EUROPE at the address mentioned in this instruction manual.
Documentation is also available at www.omron-healthcare.com.
Correct Disposal of This Product
(Waste Electrical & Electronic Equipment)
This marking shown on the product or its literature, indicates that it should not be
disposed of, with other household wastes at the end of its working life. To prevent
possible harm to the environment or human health from uncontrolled waste disposal,
please separate this product from other types of wastes and recycle it responsibly to
promote the sustainable reuse of material resources.
Household users should contact either the retailer where they purchased this product,
or their local government office, for details of where and how they can return this item
for environmentally safe recycling.
Business users should contact their supplier and check the terms and conditions of the
purchase contract. This product should not be mixed with other commercial wastes for
disposal.
TM
Baffle
Aerosol
Medication
Channel
Medication
Aerosol
Nozzle
Medication
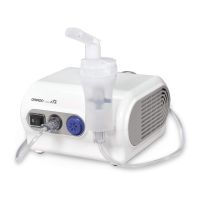
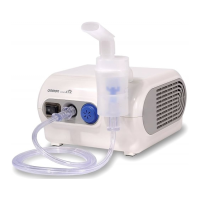
Rear View
Power Cord
Ventilation Slots
Power Plug
Ventilation Slots
Power Switch
Air Filter Cover
*Air filter inside
Air Tube
Connector
Nebulizer Kit Holder
Inhalation Top
Medication Tank
Compressor Nebulizer
Model
NE-C900
Instruction Manual
Compressed Air
Compressor (Main Unit) x 1
Nebulizer Kit x 1 Nosepiece x 1Mouthpiece x 1
Adult Mask (PVC) x 1
Child Mask (PVC) x 1
Air Tube
(PVC, 200cm) x 1
Instruction Manual
Warranty card
Spare Air Filters x 5
IM-NE-C900-E-EN-03-11/2012
1675345-3C






 Loading...
Loading...