150_OPMAN_GB Version 10 © Xstrahl Ltd. All Rights Reserved
15/01/2020
150_OPMAN_GB Version 10 ©2019
This document and all accompanying documents have been drafted
in the English language.
All manufacturer tradenames and trademarks appearing in this
document are hereby acknowledged.
Not all documents referred to in this document are part of the scope
of delivery for the equipment. Xstrahl reserves the right to determine
the documents delivered with the product.
Compatibility/Contra
indications
Xstrahl X-Ray Therapy Systems must be used only in combination
with components expressly recognised by Xstrahl as compatible with
Xstrahl X-Ray Therapy Systems. Before using any equipment or
component not supplied by Xstrahl, consult Xstrahl for advice on
compatibility.
The use of components other than those specified by Xstrahl may
affect electromagnetic compatibility (EMC) performance and result
in increased emissions or decreased immunity of the equipment.
Modification of
Equipment
Changes and/or additions to Xstrahl X-Ray Therapy Systems must
be performed only by persons expressly authorised by Xstrahl. Such
changes must comply with best engineering practice and all
applicable laws and regulations within the jurisdiction.
Any modification during the service life of the equipment requires
evaluation to the requirements of EN60601-1 and EN60601-2-8.
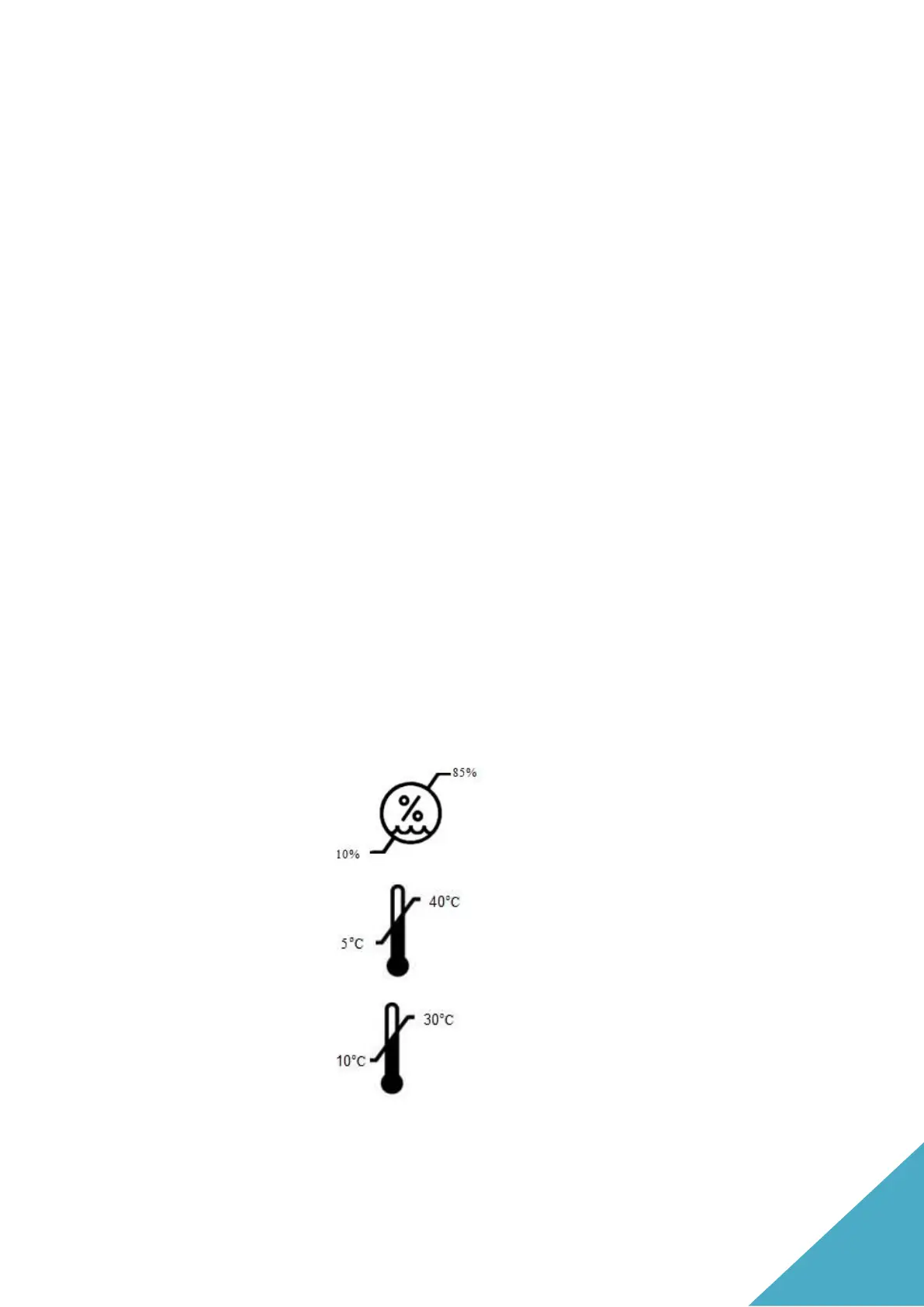
Xstrahl systems are designed to be operated and stored within the
following environmental conditions:

 Loading...
Loading...