164
e l e C t r I C a l s y s t e m s - H o u s e - 8
WARNING
Liquid lead acid batteries produce
hydrogen gas while charging. This
is highly explosive. DO NOT smoke
around batteries and keep all sources
of ignition or flames away from
batteries. The hydrogen gas may
explode resulting in fire, personal
injury, property damage or death.
Testing the Battery
A battery can be tested and monitored
several ways. The Systems Control Center,
located in the hallway area, shows the status of
the house batteries.
Checking the Electrolyte Solution:
The most efcient way
of testing the batteries is
to check the electrolyte
solution. The only way to
test a battery’s electrolyte
solution is with a hydrometer.
Many styles are available,
from types with cylinder
graduation (shown here) to
types with oating balls. Hydrometers can
be purchased from most auto parts stores.
The hydrometer tests the battery’s electrolyte
solution which is measured in specic gravity.
Distilled water has a specic assigned gravity
of 1,000. The hydrometer is calibrated to this
mark. Pure sulfuric acid has a specic gravity
reading of 1,840. The acid is 1.84 times
heavier than water. The electrolyte solution is
about 64% water to 36% acid (fully charged
battery). Hydrometers with cylinder graduation
are graphed and the exact state of specic
gravity can be determined.
Temperature and recent battery activity
(charging or discharging) affect the
hydrometer readings. It is best to check the
battery when it has been “at rest” for at least
three hours, although readings taken at other
times will give a “ballpark” gure. When using
the hydrometer, draw the
electrolyte solution up
into the tube. Allow the
hydrometer to attain the
same temperature as the
electrolyte solution. Note
the reading for that cell.
Complete the same test
for the rest of the cells on
that battery bank.
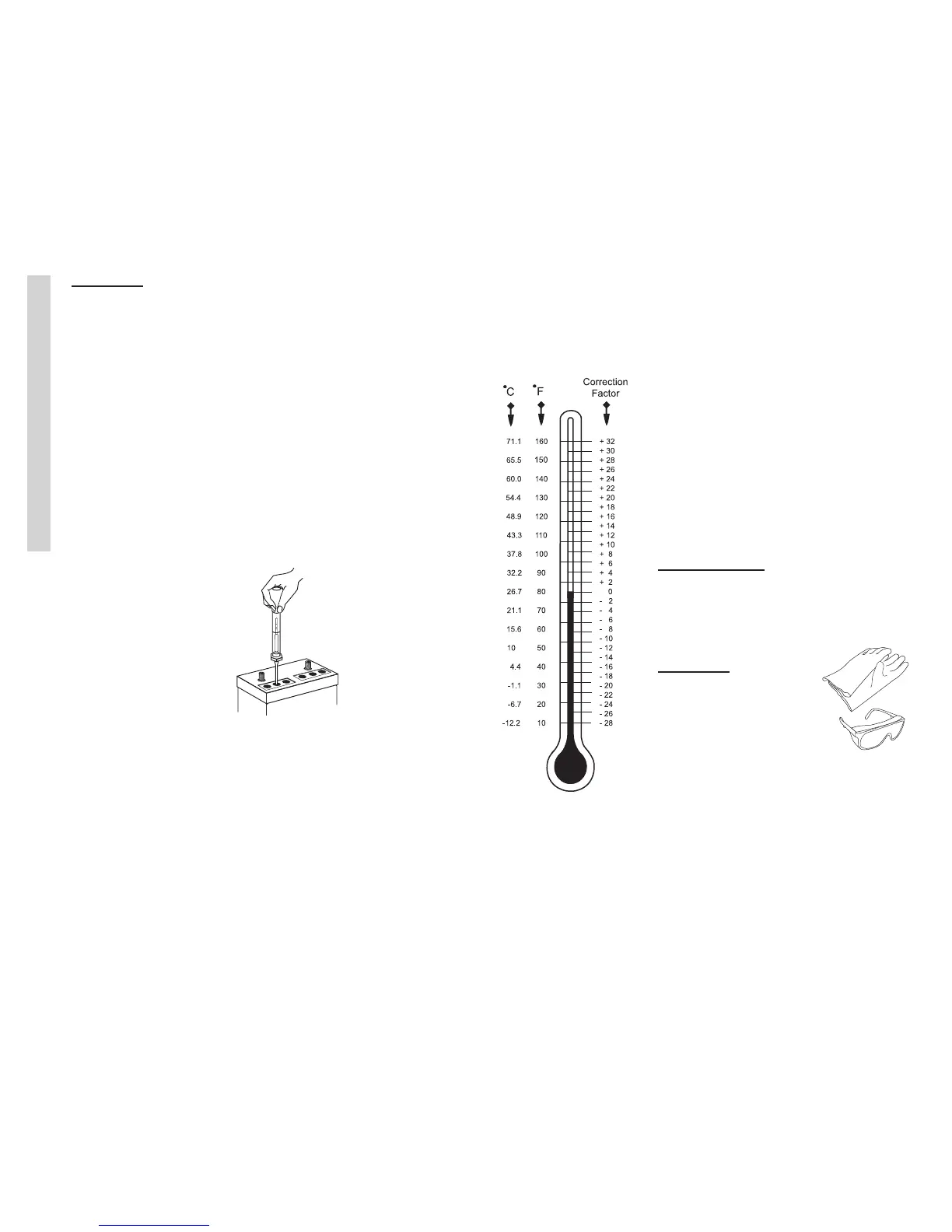
The hydrometer is
calibrated at 80° F.
Temperature affects
the hydrometer
readings. The higher the
electrolyte temperature,
the higher the specic
gravity reading. The
lower the temperature,
the lower the specic
gravity reading. Add or
subtract four points for
each 10° variance from
the 80° F chart. Readings
between cells should not
vary more than 50 points.
If one cell in a particular
battery bank being tested is at a 50% state of
charge while the others are indicating a full
charge, charge only that battery to see if the
low cell will come up. At the same time, do
not over charge the “healthy” cells.
If the low cell does not come up after
charging, this battery can damage the rest
of the battery bank and should be replaced.
An accurate digital Volt meter + - .5% will
also give an indicator of the battery’s state of
charge.
Placing a load on the Battery:
Another test that can be performed is to
place a specic load on the battery for a
predetermined length of time equal to that
particular battery’s rating. This machine is
usually an adjustable carbon pile that can vary
the load being applied to the batteries while
monitoring voltage to see if they will perform
to their specic rated capacities.
INFORMATION
See the temperature correction chart
for temperature compensation. Liquid
levels should be even between the cells
of the battery being tested as it will
affect the accuracy of the test.
WARNING
Sulfuric acid in the
batteries can cause severe
injury or death. Sulfuric
acid can cause permanent
damage to eyes burn
skin and eat holes in
clothing. Always wear splash-proof
safety goggles when working around
the battery. If the battery electrolyte
is splashed in the eyes, or on skin,
immediately flush the affected area
for 15 minutes with large quantities
of clean water. In case of eye contact,
seek immediate medical aid. Never
add acid to a battery once the battery
has been placed in service. Doing so
may result in hazardous splattering of
electrolyte.
Temperature
Correction Chart
 Loading...
Loading...