3IM-5025 REV E
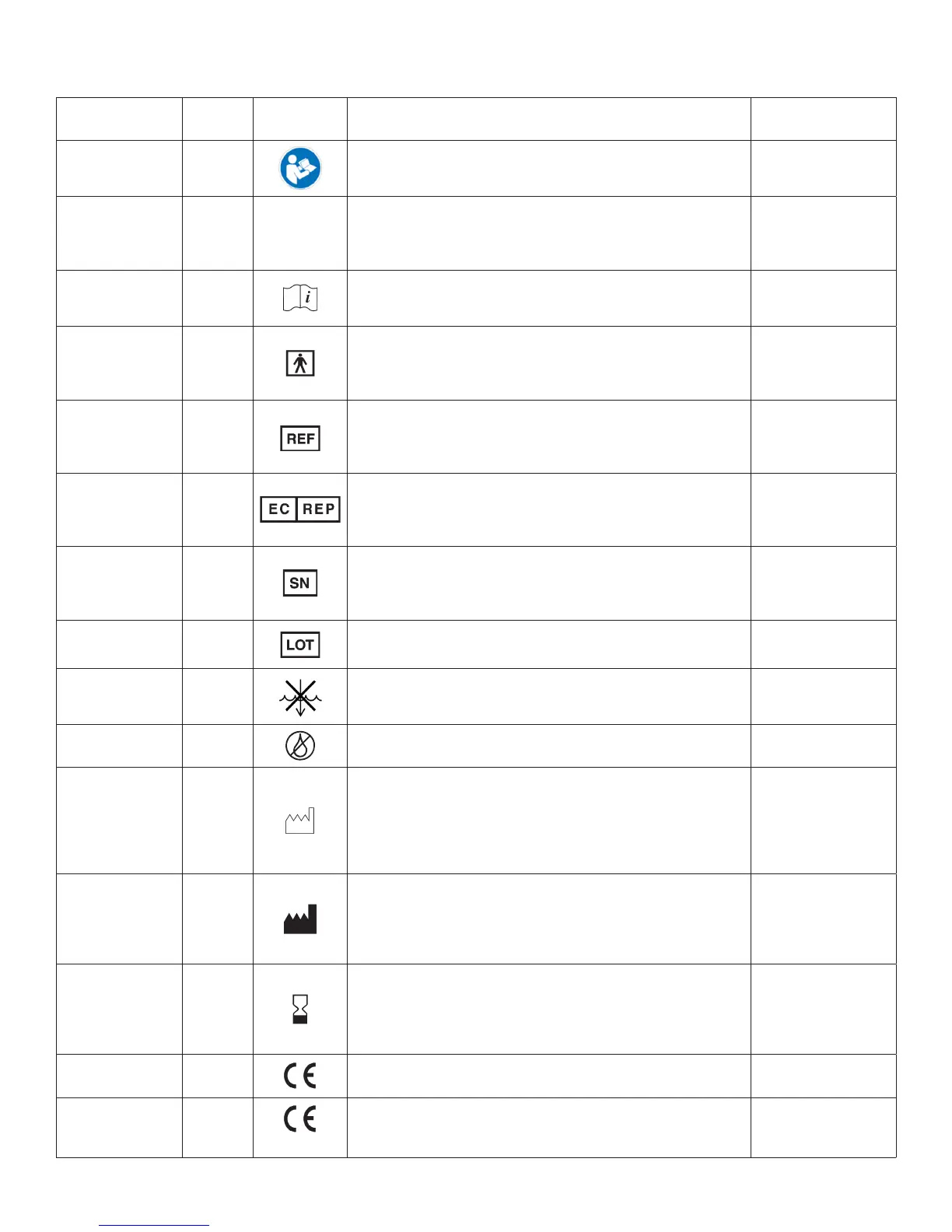
Symbol Denition
Name
Ref#
(ISO 7000)
2
Symbol Description Use Standard
Refer to Instruction
Manual / Booklet
ISO-7010
M002
• Indicates a MANDATORY action for the user to consult the In-
structions For Use (IFU).
• Symbol must be blue, as shown.
IEC 60601-1:2005
1
Caution
0434A /
0434B
C
Indicates the need for the user to consult the Instructions For Use
(IFU) for important cautionary information such as
warnings and precautions that cannot, for a variety of reasons, be
presented on the device itself.
ISO 15223-1:2012
1
Consult
Instructions For
Use (IFU)
1641
Indicates the need for the user to consult the Instructions For Use
(IFU). Not required in conjunction with the Caution
symbol, if applicable.
ISO 15223-1:2012
1
Type BF
Applied Part
5333
Indicates a medical device complying with the specied require-
ments of IEC 60601-1 to provide a higher degree of protection
against electric shock than that provided by
Type B Applied Parts.
IEC 60601-1:2005
REF (Catalog #) 2493
• Indicates the manufacturer’s catalog number so that the medical
device can be identied.
• Per EN980:2008, the REF symbol may be used without
surrounding box.
ISO 15223-1:2012
1
Authorized
Representative
in the European
Community
N/A
Indicates the authorized representative in the European
Community. This symbol shall be accompanied by the name and
address of the authorized representative, adjacent to the symbol.
ISO 15223-1:2012
1
Serial # 2498
• Indicates the manufacturer’s serial number so that a
specic medical device can be identied.
• Per EN980:2008, the SN symbol may be used without
surrounding box.
ISO 15223-1:2012
1
Lot / Batch Code 2492
Indicates the manufacturer’s batch code so that the batch or lot
can be identied.
ISO 15223-1:2012
1
Do Not Immerse in
any Liquid
5995 Indicates a medical device that is not to be immersed in any liquid. IEC 60335-2-15
Do Not Lubricate N/A Indicates a medical device that is not to be lubricated. N/A
Date of
Manufacture
2497
• Indicates the date when the medical device was
manufactured. The date is expressed as YYYY-MM (e.g. 2015-11)
or YYYY-MM-DD (e.g. 2015-11-29).
• If the symbol is lled (see Manufacturer symbol), both
the date of manufacture and the name/address of the manufac-
turer may be combined in one symbol.
ISO 15223-1:2012
1
Manufacturer 3082
• Indicates the medical device manufacturer. This symbol shall be
accompanied by the name and address of the manufacturer. The
date of manufacture may be combined with this symbol.
• When using MicroAire as the manufacturer, use the MicroAire
LLC symbol.
ISO 15223-1:2012
1
Use-By Date 2607
Indicates the date after which the medical device is not to be used.
This symbol shall be accompanied by a date to indicate that the
medical device should not be used after the end of the month
shown. The date is expressed as YYYY-MM (e.g. 2015-11) or YYYY-
MM-DD (e.g. 2015-11-29).
ISO 15223-1:2012
1
CE Mark for EU
Class I Products
N/A European Conformity Mark
Council Directive
93/42/EEC
CE Mark for EU
Class IIa and Higher
Products
N/A
0086
2013
European Conformity Mark
0086 = Notied Body Number
2013 = Year of CE Approval
Council Directive
93/42/EEC
 Loading...
Loading...