OKRA OKRA
94
95
Each OKRA Pro control solution is provided as a buered, stabilized, aqueous solution
of D-glucose containing a preservative and other non-reactive ingredient.
Level 1 0.04 wt% glucose 99.96 wt% non-reactive ingredients
Level 2 0.12 wt% glucose 99.88 wt% non-reactive ingredients
Level 3 0.20 wt% glucose 99.80 wt% non-reactive ingredients
The control solution is for In-Vitro Diagnostic use only. This means that it is only used
for testing outside of the body.
- The control solution is made for the OKRA Pro glucose monitoring system. Do not
use any other brand of control solutions.
- Check the expiration date on the bottle. Do not use if expired.
- Do not swallow. Not for human or animal consumption.
At room temperature, control solution test results will fall within the expected range
printed on the test strip bottle.
Reagents
Precautions
Expected Results
If you are using the Level 2 control solution, according to this particular vial of strips,
your meter should show a number between 80 and 120.
If your control solution test falls out of range, please follow the next steps before
contacting customer support:
· Check the expiration dates on all the products you are using. Be sure you are using
OKRA Pro control solution.
· Try another control solution test.
If this test falls out of range, try another control solution test with a new unopened
vial of strips.
· After following the appropriate steps and the control test still falls out of range, do
not perform a glucose test. Please call customer support toll free: (833-977-1339)
* Caution: The ranges in the picture above are examples only.
Please refer to your test strip vial label for the exact ranges.
For example only
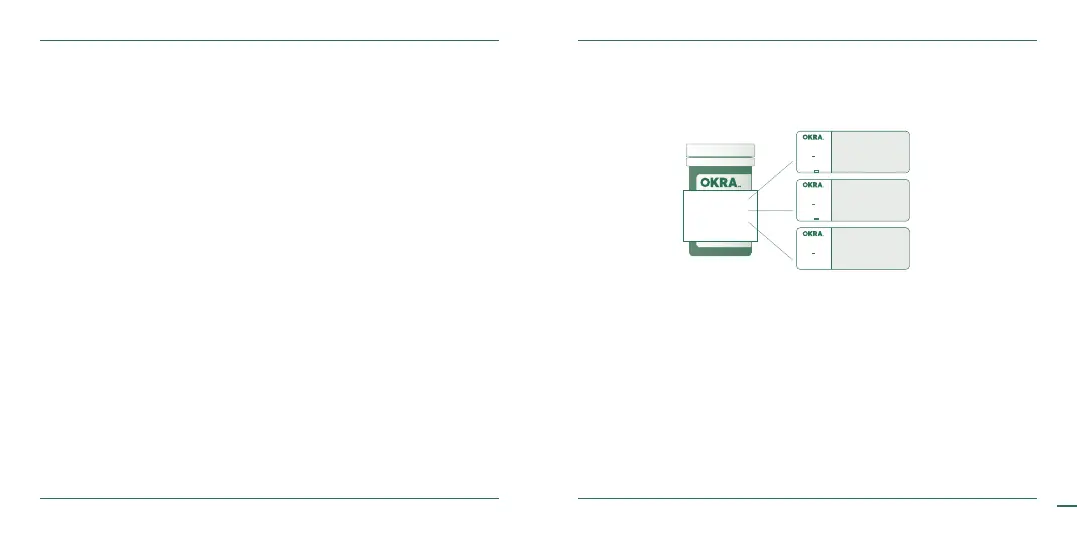
CODE
125
Level 1 35-65
Level 2 80-120
Level 3 255-345
Blood Glucose Control Solution
Level 1
3.5mL
ISC02629
INFCL003SFA
Made in Korea
Rev.2020-11-01
Level 2
Level 3
REF
3.5mL
ISC02655
INFCN001LF
Made in Korea
Rev.2020-10-01
REF
3.5mL
ISC02631
INFCL003SFA
Made in Korea
Rev.2020-11-01
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
Blood Glucose Control Solution
Blood Glucose Control Solution
Blood Glucose Control Solution
Level 1
3.5mL
ISC02629
INFCL003SFA
Made in Korea
Rev.2020-11-01
Level 2
Level 3
REF
3.5mL
ISC02655
INFCN001LF
Made in Korea
Rev.2020-10-01
REF
3.5mL
ISC02631
INFCL003SFA
Made in Korea
Rev.2020-11-01
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
Blood Glucose Control Solution
Blood Glucose Control Solution
Blood Glucose Control Solution
Level 1
3.5mL
ISC02629
INFCL003SFA
Made in Korea
Rev.2020-11-01
Level 2
Level 3
REF
3.5mL
ISC02655
INFCN001LF
Made in Korea
Rev.2020-10-01
REF
3.5mL
ISC02631
INFCL003SFA
Made in Korea
Rev.2020-11-01
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
LOT :
Expiry date :
Manufacture Date :
Discard date :
WARNING :
Close immediately after use. Read Instructions.
Shake well.
Store between 46-86° F(8-30°C).
Do not Refrigerate.
Do not Freeze.
Discard 3 months after opening.
In-Vitro Diagnostic Use.
DISTRIBUTED BY CGETC Inc.
CITY OF INDUSTRY, CALIFORNIA 91748
www.okra.care
Blood Glucose Control Solution
Blood Glucose Control Solution
 Loading...
Loading...