1
Product Description
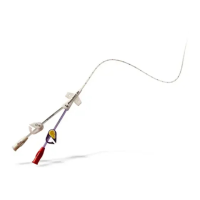
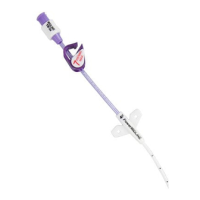
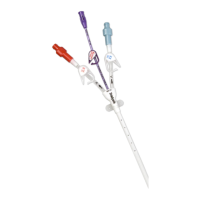
A family of peripherally inserted central catheters made from specially-formulated and processed medical grade materials.
PowerPICC® Provena™ Catheters have a kink resistant, reverse tapered design. All lumens of the 3F Single Lumen and
4F Dual Lumen catheters are appropriate for use with Central Venous Pressure (CVP) monitoring. Catheters are packaged in a kit
with accessories for reliable long (greater than 30 days) or short (less than 30 days) term vascular access, as clinically indicated.
Sterilized by ethylene oxide. Do not re-sterilize.
Indications
The PowerPICC® Provena™ Catheter is indicated for short or long term peripheral access to the central venous system for
intravenous therapy, blood sampling, power injection of contrast media, and allows for central venous pressure monitoring. For
central venous pressure monitoring, it is recommended that a catheter lumen of 20 gauge or larger be used.
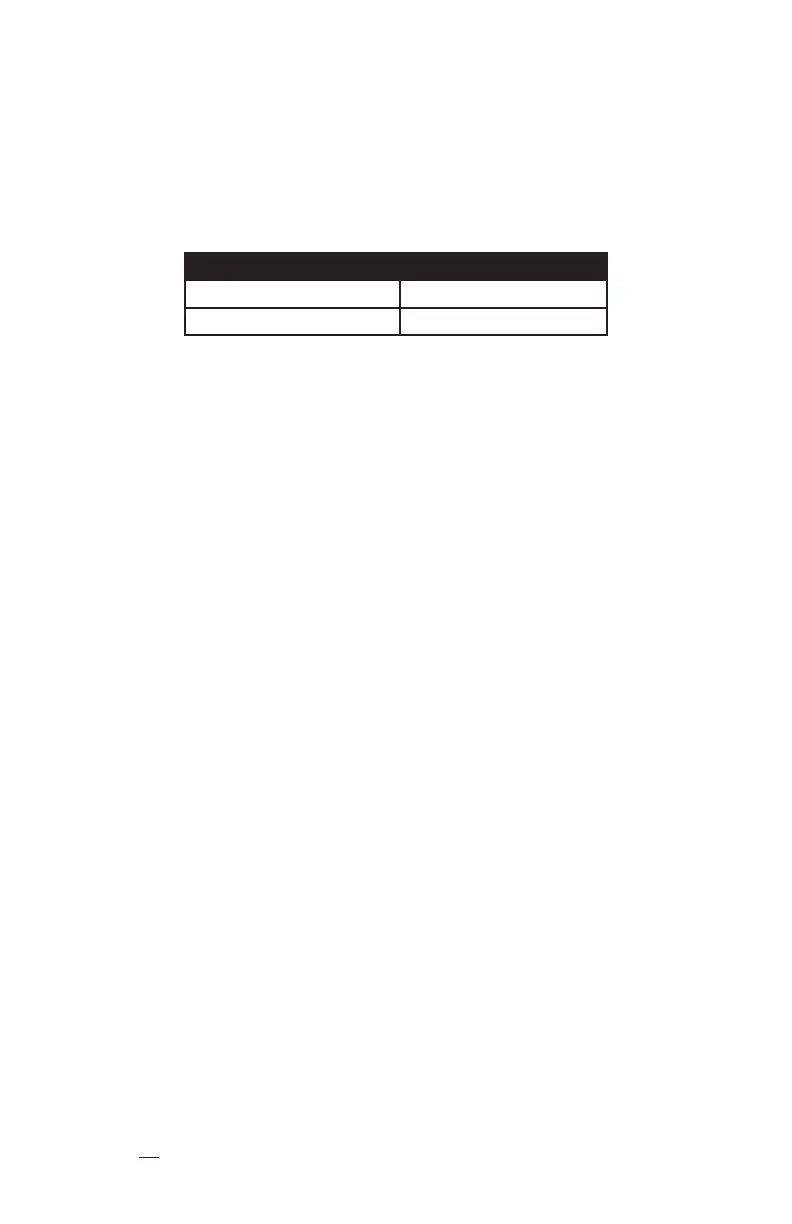
Catheter Size Maximum Flow Rate
3 F Single Lumen 3 mL / sec
4 F Dual Lumen 5 mL / sec
Contraindications
The device is contraindicated whenever:
• The presence of device-related infection, bacteremia, or septicemia is known or suspected.
• The patient’s body size is insucient to accommodate the size of the implanted device.
• The patient is known or is suspected to be allergic to materials contained in the device.
• Past irradiation of prospective insertion site.
• Previous episodes of venous thrombosis or vascular surgical procedures at the prospective placement site.
• Local tissue factors will prevent proper device stabilization and/or access.
Warnings
General Warnings
• When using alcohol or alcohol-containing antiseptics with polyurethane PICCs, care should be taken to avoid prolonged
or excessive contact. Solutions should be allowed to completely dry before applying an occlusive dressing. Chlorhexidine
gluconate and/or povidone iodine are the suggested antiseptics to use.
• Alcohol should not be used to lock, soak or declot polyurethane PICCs because alcohol is known to degrade polyurethane
catheters over time with repeated and prolonged exposure.
• Do not use the catheter if there is any evidence of mechanical damage or leaking. Damage to the catheter may lead to
rupture, fragmentation, possible embolism and surgical removal.
• If signs of extravasation exist, discontinue injections. Begin appropriate medical intervention immediately.
• Do not wipe the catheter with acetone-based solutions, tincture of iodine or polyethylene glycol-containing ointments.
These can damage the polyurethane material if used over time.
• Intended for single use. DO NOT REUSE. Reuse and/or repackaging may create a risk of patient or user infection,
compromise the structural integrity and/or essential material and design characteristics of the device, which may lead to
device failure, and/or lead to injury, illness or death of the patient.
• The uid level in the catheter will drop if the catheter connector is held above the level of the patient’s heart and opened to
air. To help prevent a drop in the uid level (allowing air entry) while changing injection caps, hold the connector below the
level of the patient’s heart before removing the injection cap.
• Central Venous Pressure (CVP) monitoring should always be used in conjunction with other patient assessment metrics
when evaluating cardiac function.
Placement Warnings
• If the artery is entered, withdraw the needle and apply manual pressure for several minutes.
• Place a nger over the orice of the sheath to minimize blood loss and risk of air aspiration. The risk of air embolism is
reduced by performing this part of the procedure with the patient performing the Valsalva maneuver until the catheter is
inserted into the sheath.
• This is not a right atrium catheter. Avoid positioning the catheter tip in the right atrium. Placement or migration of the
catheter tip into the right atrium may cause cardiac arrhythmia, myocardial erosion or cardiac tamponade. The risk of these
complications may be more likely in neonatal patients.
• Ensure that the stylet tip does not extend beyond the trimmed end of the catheter. Extension of the stylet tip beyond the
catheter end may result in vessel damage, stylet damage, dicult removal, stylet tip separation, potential embolism and/or
risk of patient injury.
Power Injection Warnings
• Exceeding the maximum power injection ow rate or setting the power injector pressure limit above 300 psi may result in
catheter failure and/or catheter tip displacement.
• Failure to ensure patency of the catheter prior to power injection studies may result in catheter failure.
• Failure to warm contrast media to body temperature prior to power injection may result in catheter failure.
• Use of lumens not marked “Power Injectable” for power injection of contrast media may cause failure of the catheter.

 Loading...
Loading...