1.
INTRODUCTION
Carbon monoxide (chemical symbol CO) is a colourless, odourless, but extremely
poisonous gas that is present in the exhaust gas of petrol-engined vehicles. The amount
of carbon monoxide in the exhaust gas is an accurate indicator of the
airlfuel mixture
strength being supplied to the engine, and for this reason motor manufacturers use the
measurement of carbon monoxide in the engine exhaust as the recommended method
for setting the
airlfuel mixture strength on carburettors and fuel injection systems. The
recommended
percentageof carbon monoxide in the exhaust at engine idle~(i.e. tickover
speed) is usually specified in the engine maintenance handbook for each vehicle.
Manufacturers typically specify a CO level somewhere within the
range0.5% to 3.5%
by volume, and
often give an upper and lower limit for the recommended setting, for
example, a manufacturer may specify 0.5% to 1.5% CO. Alternatively, the data may
be
given in the for 2%0+ .5% CO (which means between 1.5% and 2.5%). Less commonly
(and less exactly) a manufacturer may simply specify a maximum limit
e.g. below 3.5%
CO.
As can be seen, carbon monoxide only amounts to a relatively small percentage of
the total
volumeof exhaust gas. The bulk of exhaust gas comprises nitrogen
(N4,
carbon
dioxide
(C02), water vapour (H20). Hydrogen (HZ) is also present, particularly in
association with carbon monoxide. Oxygen (02) can be present either due to a weak
mixture, or due to engine misfiring. Very small amounts of other substances are also
present in exhaust gas, such as unburnt or partially burnt fuel (generally referred to as
hydrocarbons) and also some oxides of nitrogen. The way that the composition of
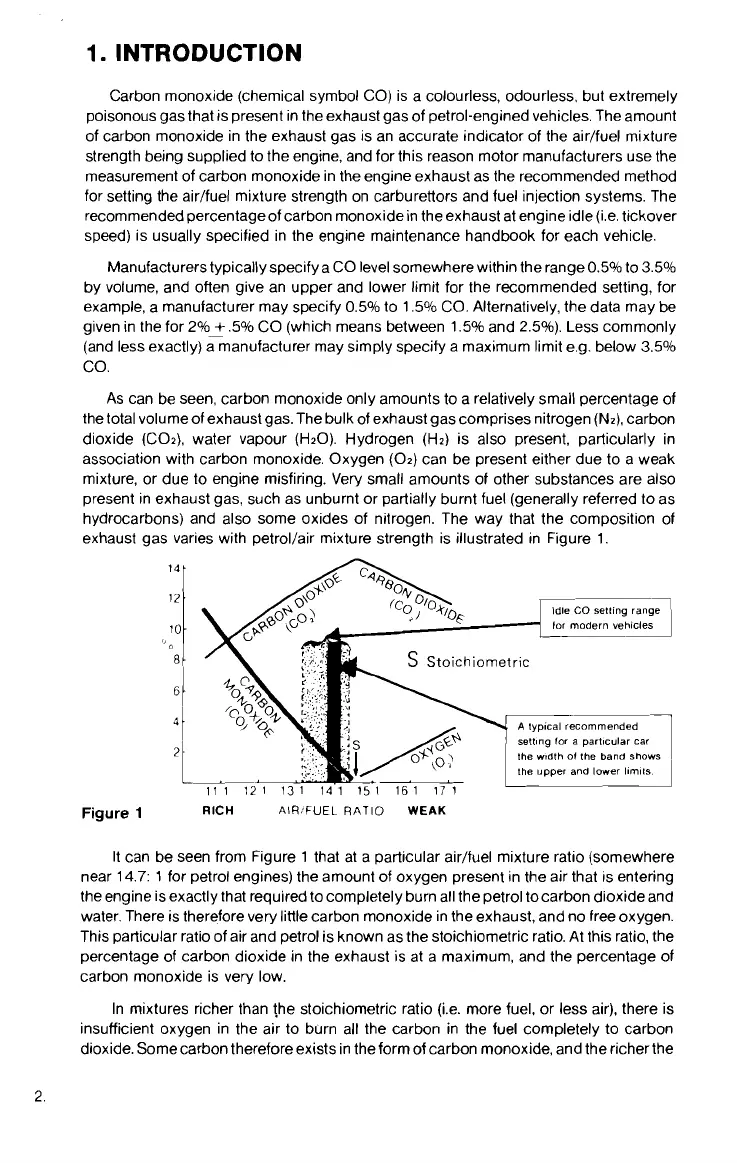
exhaust gas varies with
petrollair mixture strength is illustrated in Figure 1.
1
Idle
GO
setting
rang7
lor
modern veh~cles
A
lyplcal recommended
settong tor
a
particular
car
the
wtdth of the band shows
,
the upper and lower bmlls
Figure
1
RICH
AIRjFUEL RATIO
WEAK
It can be seen from Figure 1 that at a particular airlfuel mixture ratio (somewhere
near 14.7:
1
for petrol engines) the amount of oxygen present in the air that is entering
the engine is exactly that required to completely burn all the petrol
tocarbon dioxide and
water. There is therefore very little carbon monoxide in the exhaust, and no free oxygen.
This particular ratio of air and petrol is known as the stoichiornetric ratio. At this ratio, the
percentage of carbon dioxide in the exhaust is at a maximum, and the percentage of
carbon monoxide is very low.
In mixtures richer than
!he stoichiometric ratio (i.e. more fuel, or less air), there is
insufficient oxygen in the air to burn all the carbon in the fuel completely to carbon
dioxide. Some carbon therefore exists in the form of carbon monoxide, and the richerthe
 Loading...
Loading...

