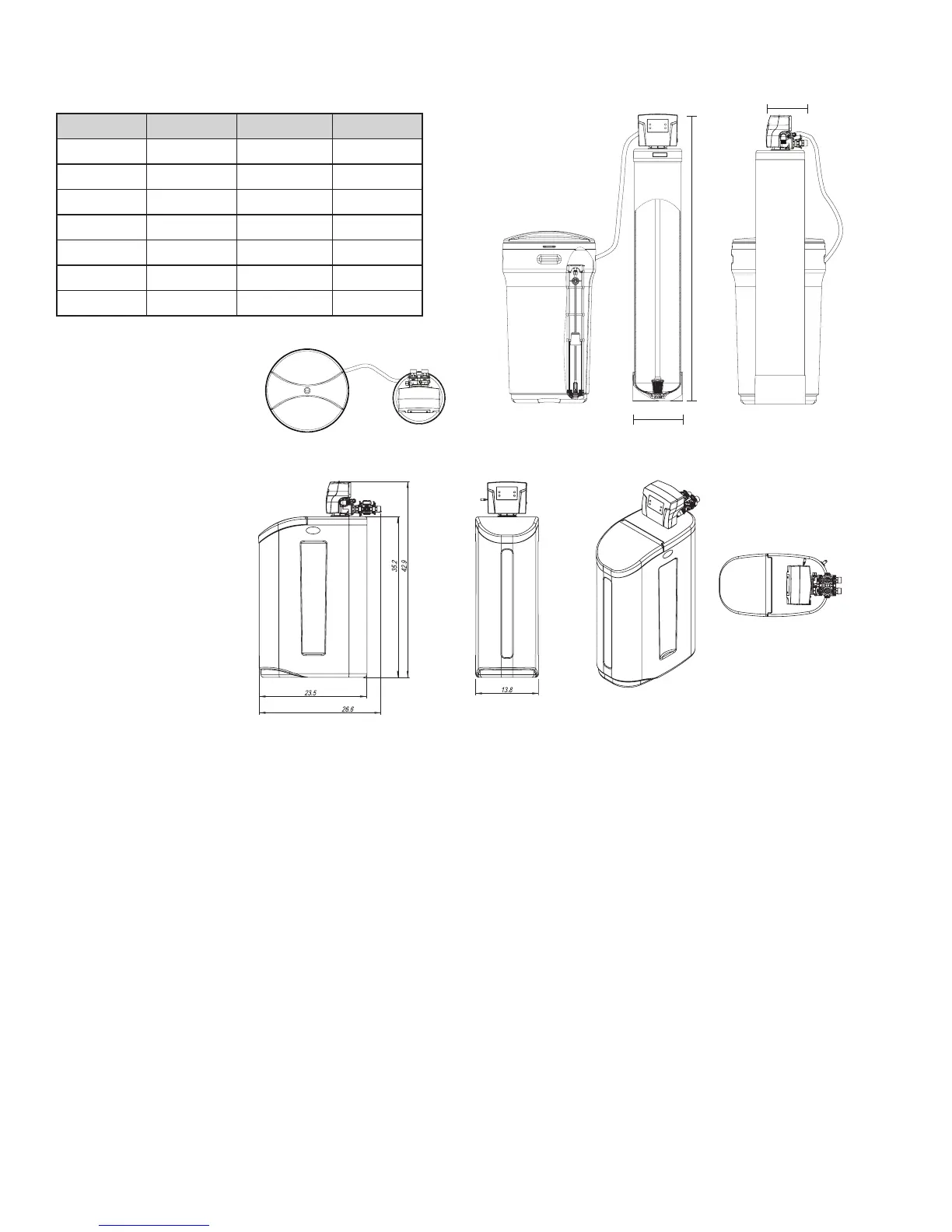
6 7
Cabinet Model
B
Twin Tank Model
A
C
Models A (Inches) B (Inches) C (Inches)
75 53” 9" 13”
100 57” 9" 13”
150 63” 10" 15”
200 61” 12" 16”
300 63” 13" 17”
400 74” 14" 18”
? 74” 16" 19”
SYSTEM DIMENSIONS
Water softeners remove hardness in the water by exchanging particles in the water, or ions. They remove hard ions the calcium and magnesium in the water by trading it
for sodium ions producing soft water. Unlike the calcium and magnesium, sodium stays dissolved in water and does not form a scale. Sodium also does not interfere with
the cleaning action of soaps. The sodium is released by a charged resin contained in the softener, this resin also traps the calcium and magnesium ions. Eventually this resin
releases all of its sodium and has filled up with other ions, so it then must be regenerated. Regeneration is accomplished by washing the resin with a salt saturated brine
solution that removes the calcium and magnesium while replenishing the sodium. This is why the softener requires a brine tank and salt. The water softener can run for days
before running out of sodium, and when it does, the sodium is replenished in only a matter of a few hours
HOW A WATER SOFTENER WORKS
 Loading...
Loading...