REF: LIB-1, LIB-3
LiNA Librata™
LiNA Medical ApS - Formervangen 5 - DK-2600 Glostrup - DENMARK - Tel: +45 4329 6666 - Fax: +45 4329 6699 - e-mail: info@lina-medical.com - www.lina-medical.com
2018-10 FV0151B
LiNA is a registered trademark of LiNA Medical in EU, US and CN
Original IFU
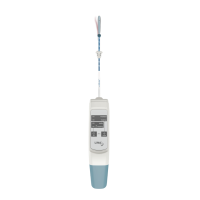
3. Insert the battery into the device and place the
device on the sterile field.
The LiNA Librata is designed with 47 minutes of
battery life, that covers one procedure only.
NOTE: Do not press the “ON”-button before all
preparations have been made (as the heating
process will then begin).
4. Conduct a pelvic examination to determine the
position of the uterus. Particular attention should
be made when sounding, dilating and inserting
the device into uterus. Insert a speculum into the
vagina and grasp with a tenaculum to bring the
uterus to a mid-plane position.
– 4a.Using a uterine sound, measure the
length of the uterus from the external os
to the fundus. Set the indicator ring on the
catheter to the measurement obtained from
the sounding. (Please note the markings on
the Librata catheter are intentionally 0.5cm
shorter than indicated. This allows a 0.5cm
gap between the balloon and fundus once
the catheter is inserted until the indicator
ring reaches the cervix).
NOTE: Due to the small diameter of the
LiNA
Librata
catheter, dilation of the cervix may not be
required. In the event that the cervix needs to be
dilated, dilate the cervix to 6mm.
5. Press the “ON” button. The device will make
a short beep sound and the “HEATING” light
will start flashing. The heating process takes
approximately 10 min.
NOTE: During the heating process the device
should remain on the sterile field and should not
be hand-held or inserted into the patient.
6. Once the fluid is heated to the correct
temperature, the “HEATING” light will stop
flashing. The device will now make a short beep
sound and the “READY” light will illuminate. The
device is now ready and
LiNA Librata
treatment
can be initiated.
NOTE: When the device is ready, the LiNA
Librata treating process needs to be started
within 30 minutes. If the treating process is not
started within this time frame, the fluid can NOT
be reheated and the procedure may therefore
be aborted.
NOTE: Please note that the device will make
a short beep sound every minute until the
treatment is started.
7. Carefully insert the catheter into the uterus
until the indicator ring touches the cervix. If
resistance is felt before the indicator ring is
touching the cervix, remove the catheter and
take a second sounding of the uterus and adjust
the indicator ring accordingly.
NOTE: Do not use excessive force when
inserting the balloon catheter.
8. When the device has been inserted correctly
into the uterus, the treating process may begin.
Press and hold the “TREAT” button for 2 seconds
and the “TREATING” light will then begin to flash.
9. The treating process takes approximately
2 minutes. During the treating process fluid will
automatically be injected and withdrawn from
the balloon a total of 5 times. Each cycle will
take approx. 25 seconds.
NOTE: Prior the beginning of the treatment
process, the device performs a short 5 second
pressure test to indicate that the balloon has not
passed outside the uterine cavity. If the device
suspects that the balloon is outside the uterine
cavity the device will abort the procedure. All
lights on the device will flash simultaneously.
A hysteroscopy should then be performed to
confirm that a perforation has not taken place. If
no perforation is revealed during hysteroscopy,
a second
LiNA Librata
device may be used.
10. When the treating process is done the
“FINALIZED” light will illuminate and the device
will make a short beep sound.
11. Remove the catheter slowly from the uterus.
NOTE: If unexpected resistance is felt during
removal of the catheter pull the “Emergency
Pull” strap on the back of the device until the
word “STOP” appears. Wait 5 seconds and
remove the catheter slowly from the uterus.
12. Dispose of the device. The battery and
the device may now be safely disposed in
accordance with local governing ordinances
and recycling plans. If desired the battery can
be removed from the device and disposed
separately.
DECLARATION OF CONFORMITY
with the
Medical Device Directive
We,
LiNA Medical ApS
Formervangen 5
2600 Glostrup
Denmark
under the sole responsibility of the manufacturer
declare that the catalogue number(s):
Product name(s) REF number(s)
LiNA Librata LIB-1
LiNA Librata LIB-3
are in conformity with the European Medical Device
Directive 93/42/EEC amended by 2007/47/EC in
effect as of
21st of March 2010 and pertinent national statutory
order.
Are in conformity with: Machinery Directive
2006/42/EC, RoHS Directive 2011/65/EU and
Standard’s: EN ISO 14971:2012; EN/IEC 60601-
1:2005+AMD1:2012; EN/IEC 60601-1-2:2014; EN/
IEC 60601-1-6:2010 +AMD1:2013; EN/IEC 60601-
2-18:2009; EN-ISO 10993-1:2009; EN ISO 10993-
10:2013; ISO 10993-11:2017; EN ISO 10993-12:2012;
EN ISO 10993-5:2009; EN 1041:2008+A1:2013;
ASTMF1980; ASTM D4169-08; EN ISO 11135:2014;
GB
DEVICE ALARMS:
Light pattern Description Reason for error User instruction
The 2 green lights are
flashing.
Device failure for
technical reasons only,
no patient safety issue is
suspected.
Dispose of the device
and battery.
Replace with a new
device if device failure
occurs before the
treatment has started.
The 2 orange lights are
flashing.
The uterine cavity is too
big to treat. The patient
cannot be treated with
the LiNA Librata
device.
Dispose the device and
battery. A hysteroscopy
might be considered
at this point to confirm
the size and shape of
the uterus to determine
alternate treatment
choices.
All lights are flashing. The balloon was unable
to meet a minimal
pressure before initiating
treatment. The balloon
may be outside the
uterine cavity.
Dispose of the device
and battery. Perform a
hysteroscopy to ensure
that a perforation of the
uterus has not occurred.
SYMBOLS USED ON PRODUCT LABELING:
CE mark with number – confirmation of
meeting EU requirements
Sterilized using ethylene oxide
Manufacturer Sterilised using irradiation
LOT
LOT Batch code Consult instructions for use
Use-by date Do not use if package is damaged
Do not reuse Type BF equipment IEC 60601
Caution
EN ISO 11137-1:2015; EN ISO 11737-1:2006; EN
ISO 11737-2:2009, EN 62304:2006/A1:2015; EN ISO
14644-1:2015; EN ISO 11607-1: 2009/A1:2014, EN
15223:2016.
CE marking 2016.
 Loading...
Loading...