LiNA Medical ApS - Formervangen 5 - DK-2600 Glostrup - DENMARK - Tel: +45 4329 6666 - Fax: +45 4329 6699 - e-mail: info@lina-medical.com - www.lina-medical.com
2018-10 FV0151B
LiNA is a registered trademark of LiNA Medical in EU, US and CN
• Do not remove the battery during the treatment
process.
• If a balloon leak is suspected during treatment,
abort the procedure by following the
instructions below in ‘To Abort the Procedure’.
• If unexpected resistance is felt during removal
of the catheter pull the “Emergency Pull”
strap on the back of the device until the word
“STOP” appears. Wait 5 seconds and remove the
catheter slowly from the uterus.
• Do not insert the balloon catheter into the
patient if there is glycerin and/or air in the
balloon.
POTENTIAL RISKS
As with any procedure in which the uterus is
instrumented certain risks exist. Hysteroscopy, and
endometrial ablation are well-known procedures.
Risks associated with the
LiNA Librata
device and/
or endometrial procedure, include but may not be
limited to the following:
• Nausea
• Vomiting
• Urinary Tract Infection (UTI)
• Laceration
• Abdominal Pain
• Thermal injury to adjacent tissue, including
cervix, vagina, vulva and/or perineum, fallopian
tubes and adjacent organs (e.g. bowel burn)
• Vaginal infection
• Endometritis
• Hematometra
• Diarrhea
• Fever
• Headache
• Abdominal distension
• Perforation of uterus or creation of a false
passage within the uterine wall
• Risks associated with hysteroscopy
• Infection or sepsis
• Complications leading to serious injury or death
PRECAUTIONS:
• The safety and effectiveness of the LiNA Librata
has not been fully evaluated in patients:
– with uterine sound >10 cm
– with uterine sound <6 cm
– with bicornuate or septate uteri or previous
endometrial resection/ablation
– who are post-menopausal
• A patient with submucousal / intramural
myomas such that the uterine cavity is
significantly distorted.
• Uterine wall perforation or the creation of a
false passage within the uterine wall can occur
during any procedure in which the uterus is
instrumented, especially in cases of severe
anteverted, retroflexed, or a laterally displaced
uterus.
• It is important that when the LiNA Librata
balloon catheter is advanced in to the uterus
that the indicator ring (set at the depth obtained
during sounding) reaches the cervix without
meeting resistance. If resistance is felt prior
to the indicator ring reaching the cervix, the
balloon catheter should be removed and a
second sounding should be performed and the
indicator ring adjusted accordingly.
• Use caution to ensure that the device is properly
positioned in the uterine cavity, ensure that
the catheter advances to the depth that was
obtained during the sounding procedure.
• It has been reported that patients with a
severe anteverted, retroflexed, or laterally
displaced uterus are at an increased risk of
uterine wall perforation during any intrauterine
manipulation. The clinician should use discretion
in patient selection.
• History of repeat low transverse Cesarean
section may increase the risk of patient injury
due to thinning of the myometrium.
• Please note that portable and mobile RF
communications equipment can affect the LiNA
Librata device.
INSTRUCTIONS FOR USE:
Read the instruction for use prior to using this
device! A thinned endometrium may enhance
the results of the Librata treatment. This can be
accomplished by timing the menstrual cycle to
the early proliferative phase, administering pre-
treatment drugs such as danocrine, birth control
pills, provera, etc. GnRH agonists should not be
used to thin the endometrium. The optimum pre-
treatment regimens have not been determined at
this time.
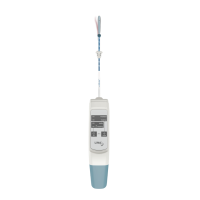
To abort the procedure:
A. Press the “ON” and “TREAT” buttons
simultaneously (“ABORT”)
B. Observe that the ‘FINALIZED’ light is
flashing and that beeps are sounding.
C. Wait 8 seconds to let the “ABORT”
feature withdraw all the fluid from
the balloon. The ‘FINALIZED’ light will
illuminate.
D. Remove the catheter slowly from the uterus.
E. In the unlikely event that the “ABORT” function
does not work, or if unexpected resistance is
felt during removal of the catheter, pull the
“Emergency Pull” strap on the back of the device
until the word “STOP” appears. This will drain the
fluid from the balloon back into device.
F. Wait 5 seconds and remove the catheter slowly
from the uterus.
NOTE: Please note that after using the “ABORT”
feature, the treatment can NOT be resumed nor can
the device be restarted.
Steps for the procedure:
1. Remove the blister tray from the environmental
pouch.
2. Carefully inspect the blister trays and Tyvek lids
for any damages prior to use. Do NOT attempt to
use the device if the sterile barrier is damaged.
Do NOT use past expiration date.
– 2a. Peel back the lids on the packaging.
Remove the device and battery following
sterile technique
GB
 Loading...
Loading...