Blood Pressure Monitor
FR IT DE PT NL
SWE
PL DKENES
28
Guidance and manufacturing declaration - electromagnetic immunity - for all EQUIPMENT
& SYSTEMS
The blood pressure monitor is intended for use in the electromagnetic environment
specied below. The customer or user must ensure that it is used in these environments.
Immunity test IEC 60601
test level
Compliance
level
Electromagnetic environment
Electrostatic
discharge (ESD)
IEC61000-4-2
±6 kV
contact
±8 kV air
±6 kV
contact
±8 kV air
Floors should be wood, concrete or
ceramic tiles. If the oor is covered with
synthetic material, the relative humidity
should be at least 30%.
Electric frequency
magnetic eld (50
Hz) IEC 61000-4-8
3 A/m 3 A/m Magnetic power frequency elds
should be at levels characteristic of a
typical hospital or commercial environ-
ment.
Guidance and Declaration of Manufacture - electromagnetic immunity - for EQUIPMENT
AND SYSTEMS other than LIFE ASSISTANCE
The blood pressure monitor is intended for use in the electromagnetic environment
specied below. The customer or user must ensure that it is used in these environments.
Immunity
test
IEC
60601
test
level
Com-
pliance
Guidance on the electromagnetic environment
IEC
61000-
4-3 RF
radiated
3 V/m
80
MHz
to 2.5
GHz
3 V/m
Portable and mobile RF communication equipment
should be used no closer to any part of the foetal bag
detector, including cables, than the recommended
separation distance, calculated from the equation
applicable to the frequency of the transmitter.
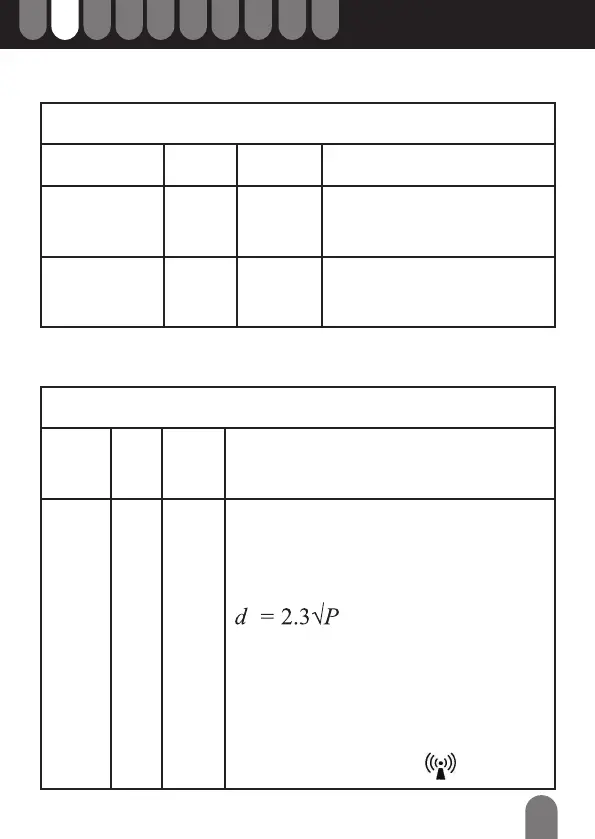
Recommended separation distance
800 MHz at 2,5 GHz
Where P is the maximum output power of the transmitter
in watts (W) according to the transmitter manufacturer
and, d is the recommended separation distance in
metres (m).
The eld extent of xed RF transmitters, as determined
by an electromagnetic site survey (a), should be less than
the compliance level in each frequency range (b).
Interference may occur in the vicinity of equipment
marked with the following symbol:
 Loading...
Loading...