33
11. Reference To Standards
Device standard: Device corresponds to the requirements of the
European standard for non-invasive Blood pressure
monitor
EN1060-1
EN1060-3
EN1060-4 – clinical investigation
IEC/EN 60601-1-11
ANSI / AAMI SP10
NIBP
IEC80601-2-30:2009 + corrigendum 2010
Electrical compatibility:
Device fulls the stipulations of the
IEC/EN 60601-1
IEC/EN 60601-1-2
The stipulations of the EU-Guidelines 93/42/EEC for Medical Products Class
IIa have been fullled.
12. Remark
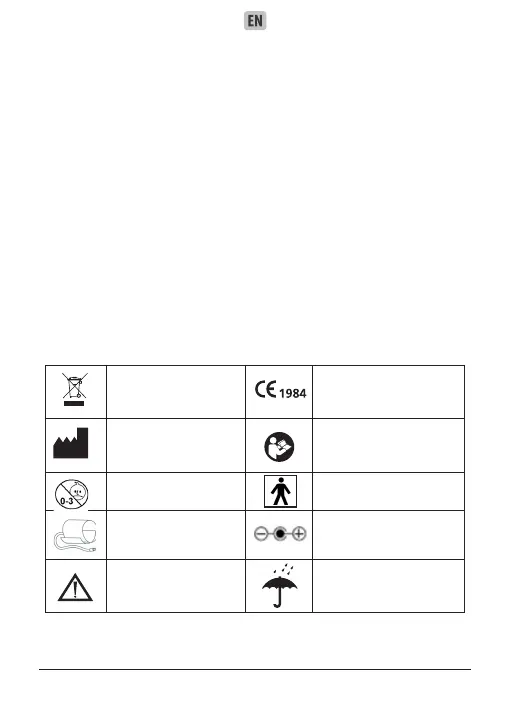
Waste Electrical and
Electronic Equipment
Directive
CE mark
Manufacturer
Read the instructions
carefully before using this
device.
Inapplicable baby Type BF equipment
Cuff Connector AC/DC Adapter
Attention consult
accompanying docu-
ments
Keep dry
 Loading...
Loading...