15
13.3 Declaration of Conformity UE
Tecnimed srl, P.le Cocchi, 12 - 21040 Vedano Olona (VA) - Italy is the manufacturer of
the VisioFocus 06400 non-contact clinical infrared thermometer.
TECNIMED guarantees that VisioFocus 06400 meets all the provisions applicable in
the Regulation (UE) 2017/745.
VisioFocus 06400 is a class IIa medical device manufactured following appro
-
priate production processes in accordance with an EN ISO 9001:2015 and EN ISO
13485:2016 certied Quality System, with GMP requirements and with the following
standards: EN 60601-1, EN 60601-1-2, EN 60601-1-6, EN 60601-1-11, EN 62366,
EN 62471, EN 62304, EN 80601-2-56, ASTM E 1965-98.
Full responsibility for the conformance of this product to the standards is assumed by
Tecnimed srl, P.le Cocchi, 12 - 21040 Vedano Olona (VA) – ITALY.
Complete Declaration of Conformity is available from the manufacturer on request:
info@tecnimed.eu
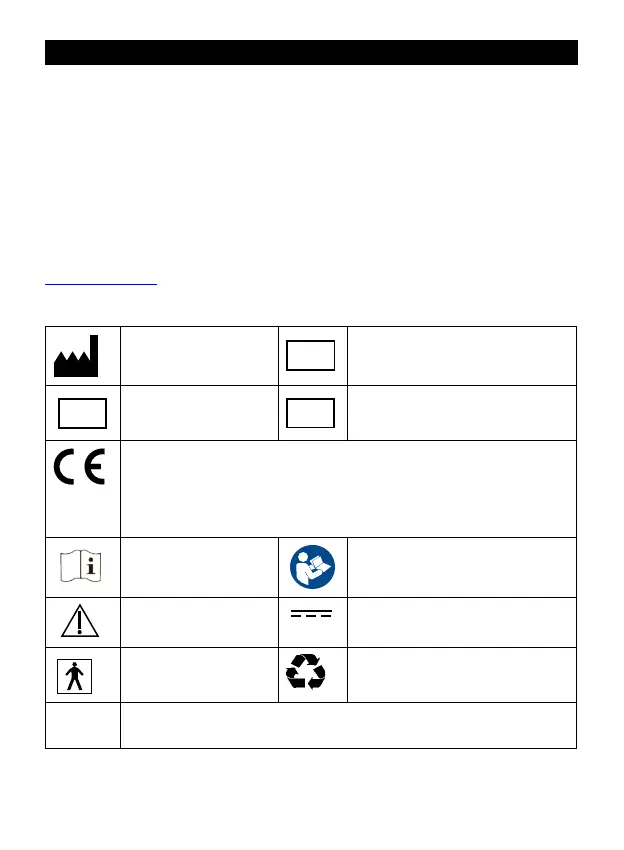
SYMBOLS on the thermometer, on its packaging or in this user manual
Manufacturer
UDI
UDI code
MD
Medical Device
SN
Serial Number
0051
European Conformity mark. The product conforms to:
- Regulation (EU) 2017/745 on medical devices. The Conformity to
the Regulation (EU) 2017/745 applies to the medical use only and it is
veried by the notied body no. 0051 (IMQ).
- 2014/30/EU directive for the non-medical use (ref. par. 4.4)
Operating
instructions
Refer to the instruction manual
CAUTION: read the
warnings
Direct current
Applied part: type BF Recyclable material
IP22
Protected against insertion of objects >12,5 mm (>0.5 inches) and
against vertical dripping water when tilted up to 15°
 Loading...
Loading...