1
3M™ Attest™ Rapid Readout Biological Indicator1295 1
ProductDescription
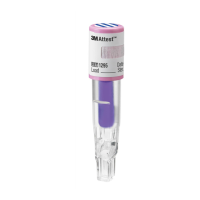
The 3M™ Attest™ Rapid Readout Biological Indicator
1295(pink cap, referred to hereinafter as the 1295BI) is
a self‑contained biological indicator specifically
designed for rapid and reliable routine monitoring of
vaporized hydrogen peroxide sterilization processes
when used in conjunction with the 3M™ Attest™
Auto‑reader 490H or the 3M™ Attest™ Auto‑reader
490having software version 4.0.0or greater
(hereinafter referred to as the 490H Auto‑reader or
490Auto‑reader having software version 4.0.0or
greater). The 1295BI is a single‑usedevice.
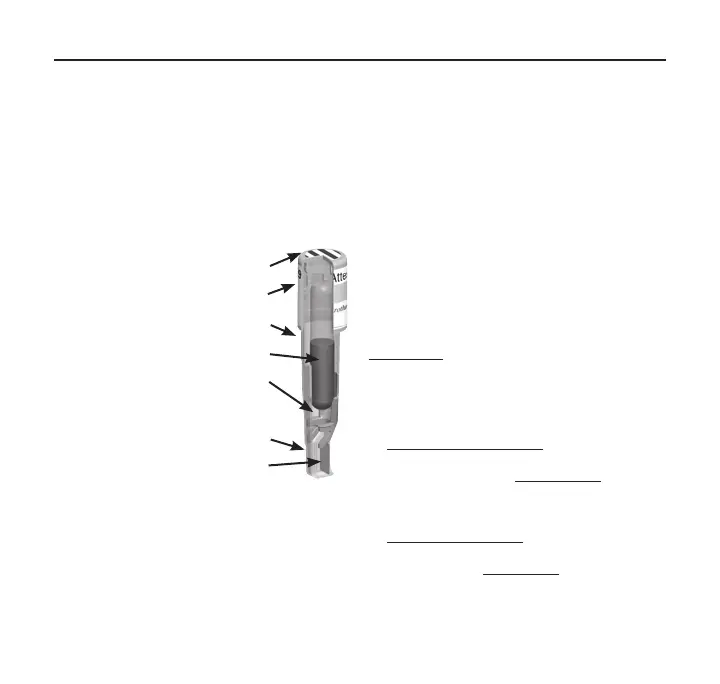
A schematic
illustrating the
design of the
1295BI is provided
in Figure 1. The
self‑contained
design includes a
carrier with spores
of Geobacillus
stearothermophilus
and a media
ampoule
containing
bacteriological
growth medium
which meets the
requirements for
growth promoting
ability specified
in ISO 11138‑1:
2017. The spore
carrier and media ampoule are contained in a plastic vial
topped with a pink cap. A chemical process indicator
printed with stripes which change in color from blue
towards pink upon exposure to vaporized hydrogen
peroxide is located on the top of thecap.
The 1295BI utilizes the ‑glucosidase enzyme system,
which is generated naturally within growing cells of
Geobacillusstearothermophilus. The ‑glucosidase
in its active state is detected by measuring the
fluorescence produced by the enzymatic hydrolysis
of a non‑fluorescent substrate, 4‑methylumbelliferyl‑
‑D‑glucoside (MUG). The resultant fluorescent
by‑product, 4‑methylumbelliferone (MU), is detected
in the Auto‑reader. The presence of fluorescence within
the specified incubation time for the 1295BI in the
Auto‑reader indicates a sterilization processfailure.
The 1295BI can also indicate the presence of
G.stearothermophilus organisms by a visual pH
color change reaction. Biochemical activity of the
G.stearothermophilus organism produces metabolic
by‑products that cause the media to change color from
purple to yellow which also indicates a sterilization
process failure. Use of this indication method is optional
and is typically restricted to specialstudies.
ReadoutTime
The rapid readout result has been correlated with a
7‑day visual pH color change result following the FDA’s
Reduced Incubation Time protocol. The time to result
is determined by the software version programmed on
the Auto‑reader.
24‑minute FluorescentResult
1295BIs incubated in a 490H or 490Auto‑reader
having software version 4.0.0or greater have a
24minute reduced incubation time result that
correlates to the 7day (168hours) visual readout
result ≥ 97% of thetime.
4‑hour FluorescentResult
1295BIs incubated in a 490H Auto‑reader having
software versions less than 4.0.0have a 4‑hour
reduced incubation time result that correlates to
the 7day (168hours) visual readout result ≥ 97% of
thetime.
PROCESS
INDICATOR
BI CAP
WITH LABEL
VIAL
MEDIA
AMPOULE
AMPOULE
CRUSHER
GROWTH CHAMBER
SPORE
CARRIER
Figure 1: Design of the
3M™Attest™ Rapid Readout
Biological Indicator 1295
 Loading...
Loading...