Description:
Base File Name:
Software:
SGS Contact:
Email:
Printer:
Printer Location:
Supplier:
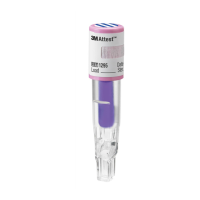
3M™ HealthCare Attest™
Rapid Readout Biological Indicator
Requester:
Katelyn Early
Reference:
1295
Die # / Doc. Size:
5" x 4.75"
Item Spec#:
34-8722-3874-5
Supersedes#:
34-8720-4945-6
Cat/Product#:
Structure#:
SS-76438
Booklet
PA: 34872238745.indd
GA:
InDesign CC 2014
PROCESS
BLACK
02.24.18 PU 1295_34872049456.indd (SGS#4599854), update GB and prep for translations kmh
02.28.18 Alts per marked up pdf kmh
03.07.18 Collect InDesign files to be sent for translations kmh
05.03.18 Update with new supplied translations. TN
05.14.18 Alt per language kmh
05.17.18 Final release. TN
2
Due to the high reliability of the fluorescent result
there is no advantage to incubating 1295BIs after the
fluorescent result has been determined by the 490H
Auto‑reader or 490Auto‑reader having software
version 4.0.0or greater andrecorded.
1295BIs meet ISO 11138‑1:2017.
Indications forUse
Use the 3M™ Attest™ Rapid Readout Biological
Indicator 1295in conjunction with the 3M™ Attest™
Auto‑reader 490H or 490Auto‑reader having software
version 4.0.0or greater as a standard method of routine
monitoring of vaporized hydrogen peroxide sterilization
processes in the following systems: AMSCO
®
V‑PRO
®
1Low Temperature Sterilization System (Lumen
cycle), AMSCO
®
V‑PRO
®
1Plus Low Temperature
Sterilization System (Lumen and Non Lumen cycles),
AMSCO
®
V‑PRO
®
maX Low Temperature Sterilization
System (Lumen, Non Lumen, and Flexible cycles),
AMSCO
®
V‑PRO
®
60Low Temperature Sterilization
System (Lumen, Non Lumen and Flexible cycles) and
in STERRAD
®
100S, STERRAD
®
NX (Standard and
Advanced cycles), STERRAD
®
100NX (Standard, Flex,
Express and Duo cycles) systems, STERRAD
®
NX with
AllClear™ Technology (Standard and Advanced cycles)
and STERRAD
®
100NX with AllClear™ Technology
(Standard, Flex, Express and Duo cycles).
Contraindications
None.
Warnings
There is a glass ampoule inside the plastic vial of the
biological indicator (BI). To avoid the risk of serious
injury from peroxideburns:
• Wear safety glasses and gloves when removing
the 1295BI from thesterilizer.
• Wear safety glasses and gloves when activating
the 1295BI.
• Handle the 1295BI by the cap when crushing
orflicking.
Residual hydrogen peroxide may be trapped within the
1295BI if the media ampoule is damaged during the
sterilization process. If a broken ampoule is observed
after processing, avoid direct contact with the 1295BI
as it may result in hydrogen peroxide burns. Follow
the disposal instructions provided at the end of
thisdocument.
Precautions
1. Do not use the 1295BI to monitor sterilization cycles
which it is not designed tochallenge:
a. Steam sterilizationcycles;
b. Dry heat sterilization cycles;or
c. Ethylene oxide sterilizationprocesses.
2. To reduce the risk associated with incorrectresults:
• Before sterilization, inspect 1295BI to verify
media ampoule is intact and process indicator
stripes are blue. Do not use any 1295BIs which
have a broken media ampoule or process
indicator stripes which are notblue.
• Do not place tape or labels on 1295BI prior to
sterilization or incubation in the Auto‑reader.
• Activate and incubate the 1295BI within 1hour
after the completion of the sterilizationcycle.
• Do not incubate a 1295BI if, after processing
and before BI activation, it is observed to have a
broken media ampoule. Retest the sterilizer with
a new biologicalindicator.
• After 1295BI activation, ensure media has flowed
to the spore growthchamber.
3. To ensure the product functions as intended
throughout the labeled shelf life, store 1295BIs in
the resealable foil pouch untiluse.
 Loading...
Loading...