AXONICS SNM SYSTEM® – Neurostimulator | Recharge-Free, Model 4101 Page 5
INTRODUCTION
This manual provides information about the Axonics Sacral Neuromodulation (SNM) System Neurostimulator (Model 4101), which is
a part of the Axonics SNM System. The Neurostimulator is connected to the Axonics Tined Lead (Model 1201 or 2201).
INDICATIONS, WARNINGS, AND PRECAUTIONS
• Refer to Indications for Use insert for indications and contraindications.
• Refer to Information for Prescribers booklet for warnings, precautions, adverse events, patient selection and clinical
summary.
• Refer to MRI guidelines for information on MRI compatibility and requirements.
These inserts and booklets can be found at www.axonics.com/eIFU.
DEVICE DESCRIPTION
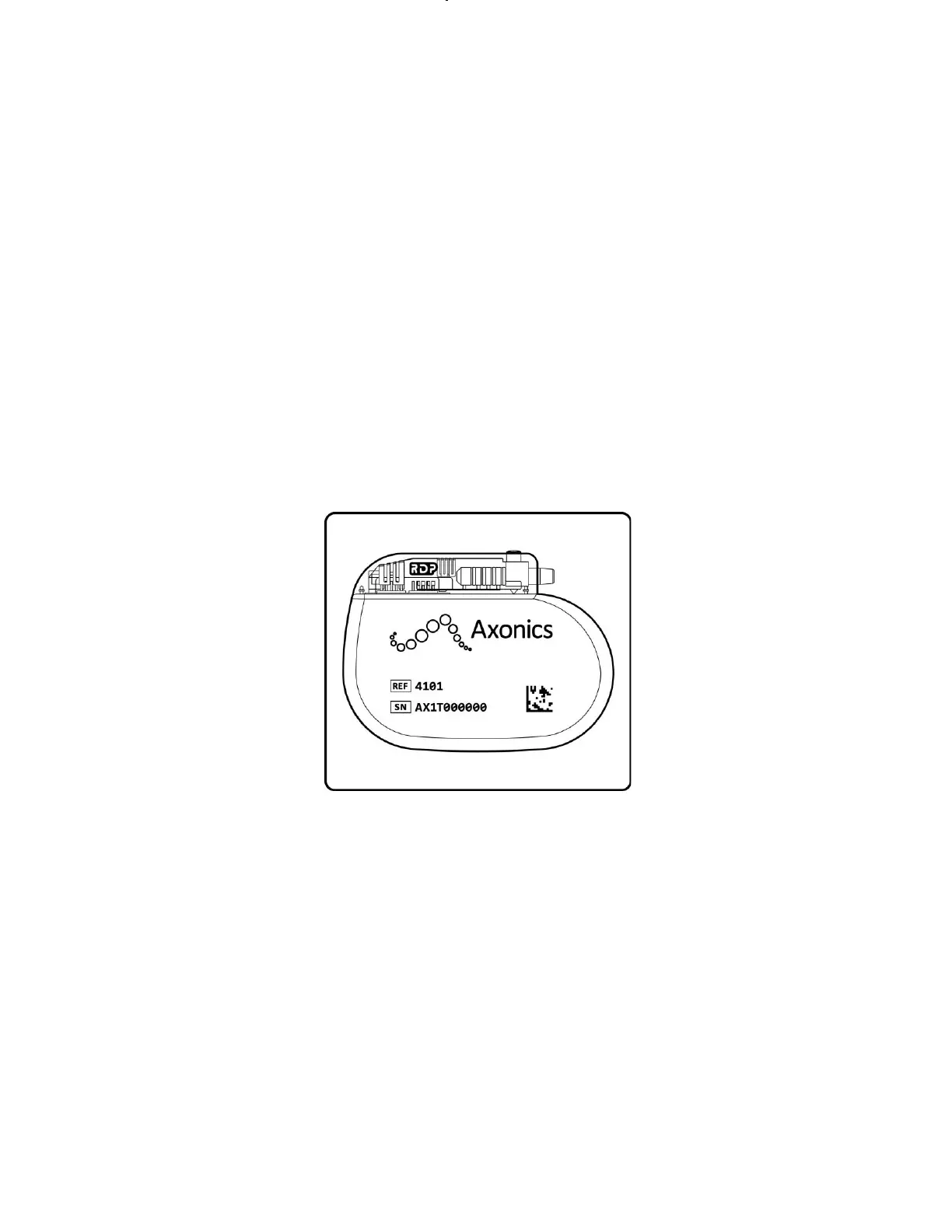
The Axonics Recharge-Free Neurostimulator (
Figure 1) is part of the Axonics SNM System. The Neurostimulator is a programmable device that is connected to the Axonics Tined
Lead, which conducts stimulation pulses to the sacral nerve.
Figure 1: Axonics Recharge-Free Neurostimulator.
Package Contents
The Neurostimulator package contains the following:
•
Neurostimulator
•
Torque wrench
•
System registration form
•
Patient identification card
•
Product Insert with Indications for Use
The contents of the inner package are STERILE.
The contents of the Neurostimulator package are intended for single use only.
System Registration Form and Patient Identification Card
The system registration form registers the device and creates a record of the device in Axonics’ implant data system.
The patient identification card is also packaged with this device. The patient should carry the identification card at all times.

 Loading...
Loading...