PAGE 4
Restrictions for the patient and the cardiac device system
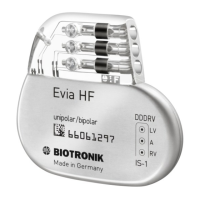
The devce system conssts of a pacemaker, ID, or RT-D wth the
respectve leads that are separately labeled MR condtonal and,
when combned, consttutes an MR condtonal devce system (See
charts on p 10)
There are no other actve or abandoned cardac mplants (eg, lead
extensons, lead adapters or abandoned leads) n the patent’s body
Other actve or passve mplants are permtted f they are dented
as MR condtonal by the manufacturer
Note An MRI scan s permtted only f the product-specc
condtons are met for all mplants and f no metal mplantable
devce longer than 5 cm s n the vcnty of a BIOTRONIK lead wthn
a dstance of less than 4 cm
The leads have been mplanted for at least 6 weeks
The devce system s mplanted pectorally
The measured pacng threshold s not above 20 V at 04 ms
pulse wdth
The devce system should be functonng normally pror to an MRI
The battery status s nether ERI nor EOS
The devce s programmed to an MRI mode before the MR scan
Conditions for MR scans with a BIOTRONIK ProMRI
®
system (continued)
 Loading...
Loading...