2 - US PN MN238 E | User Manual
VisionAire 5
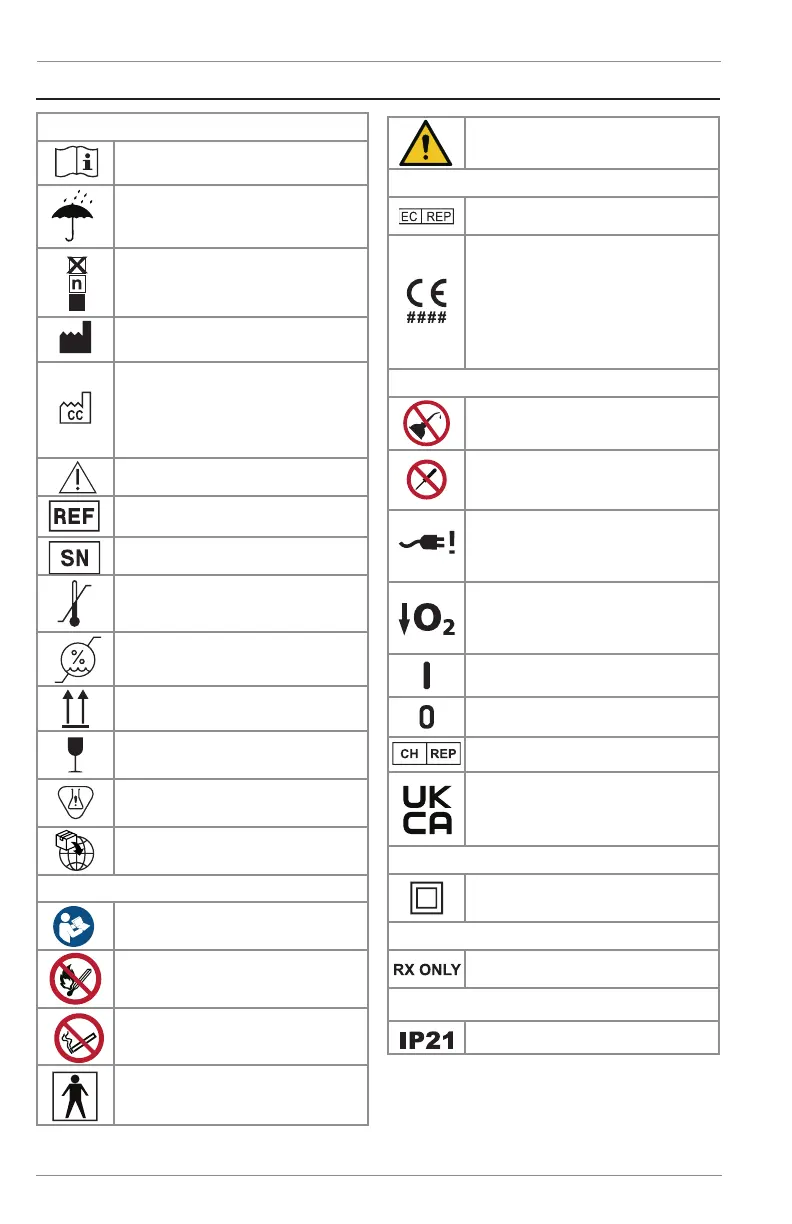
User Controls & System Status Indicators
ISO 7000
Read user’s manual before operation.
Reg. # 1641
Keep away from rain, keep dry. Reg.
# 0626
Stacking limit by number. Reg. # 2403
Name and address of manufacturer.
Reg. # 3082
The country and date of manufacture.
The “CC” identies the two letter
country code of the country of manu-
facture. The date of manufacture is in
YYYY-MM-DD format. Reg. # 6049
Caution, consult accompanying docu-
ments. Reg. # 0434A
Catalog Number. Reg. # 2493
Serial Number. Reg. # 2498
Storage or operating temperature
limitation range Reg. # 0632
Storage humidity range
Reg. # 2620
This way up. Reg. # 0623
Fragile, handle with care. Reg. #
0621
Contains hazardous substances. Reg.
# 3723
Importer. Reg. # 3725
ISO 7010
The instruction manual must be read.
Reg. # M002
Keep away from open ame, re,
sparks. Open ignition source and
smoking prohibited. Reg. # P003
Do not smoke near unit or while
operating unit. Reg. # P002
Type BF applied part (degree of
protection against electric shock).
Reg. # 5333
Warning. Reg. # W001
Council Directive 93/42/EEC
Authorized representative in the
European Community
If the product unique device identier
(UDI) label has the CE#### symbol
on it, the device complies with the
requirements of Directive 93/42/EEC
concerning medical devices. The
CE#### symbol indicates notied
body number.
Additional Symbols
Keep away from ammable materials,
oil and grease.
Do not disassemble.
When present on the device alarm
panel indicates external power inter-
ruption has been detected.
2018 Labeling: When present on the
device alarm panel indicates low ox-
ygen concentration in device output.
ON (power switch on)
OFF (power switch off)
Authorized representative in
Switzerland.
If the device bears the UKCA mark
as shown with UKCA#### indicating
the notied body number, this device
complies with UKCA regulations.
IEC 60417
Class II equipment
21 CFR 801.15
Federal law restricts this device to
sale by or on the order of a physician.
IEC 60601-1
Drip Proof Equipment - IP21
 Loading...
Loading...