en
69
8.2 Aerosol characteristics in accordance with EN 13544-1
appendix CC
The output of the nebuliser depends on the medication being
used. Consequently, the following data may not apply for medi-
cations that exist as suspensions or are very viscous. If in doubt,
ask the manufacturer of the medication concerned for advice.
Determination of particle size
Fill the INQUA
®
nebuliser with 3 ml of a 2.5% test solution and
then attach the INQUA
®
nebuliser to an impactor (NGI™ = Next
Generation Impactor™) and run for 3 minutes.
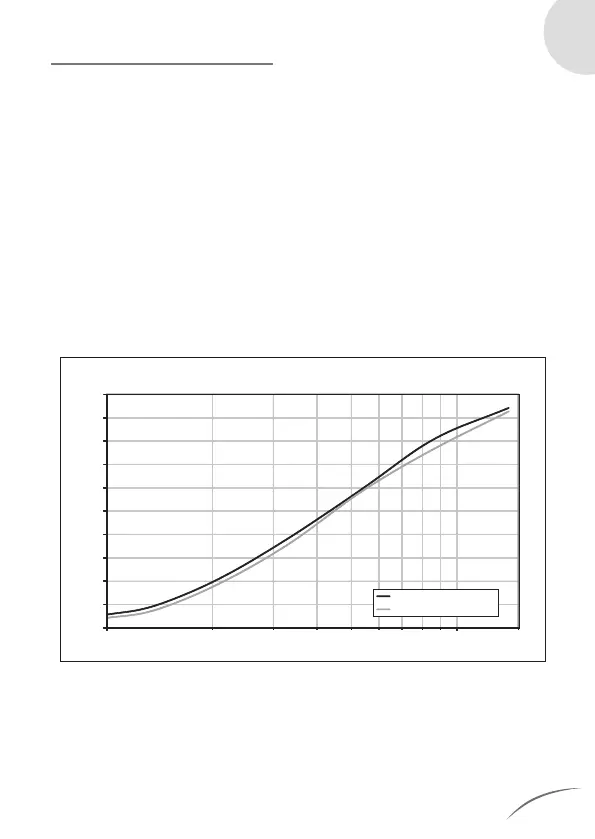
Cumulative mass distribution for the INQUA
®
nebuliser at the
minimum flow (3.0 l/min) and the maximum flow (6.0 l/min):
(Averages from each of 2 separate measurement series with 3 nebuliser/com-
pressed air combinations)
0
10
20
30
40
50
60
70
80
90
100
MMAD [µm]
1.00 10.00 15.00
Cumulative drug depostion [%]
Cumulative drug distribution [%] with 2.5% test solution
Mean Value at 3 l/min JetFlow
Mean Value at 6 l/min JetFlow
 Loading...
Loading...