INTRODUCTION
1.4
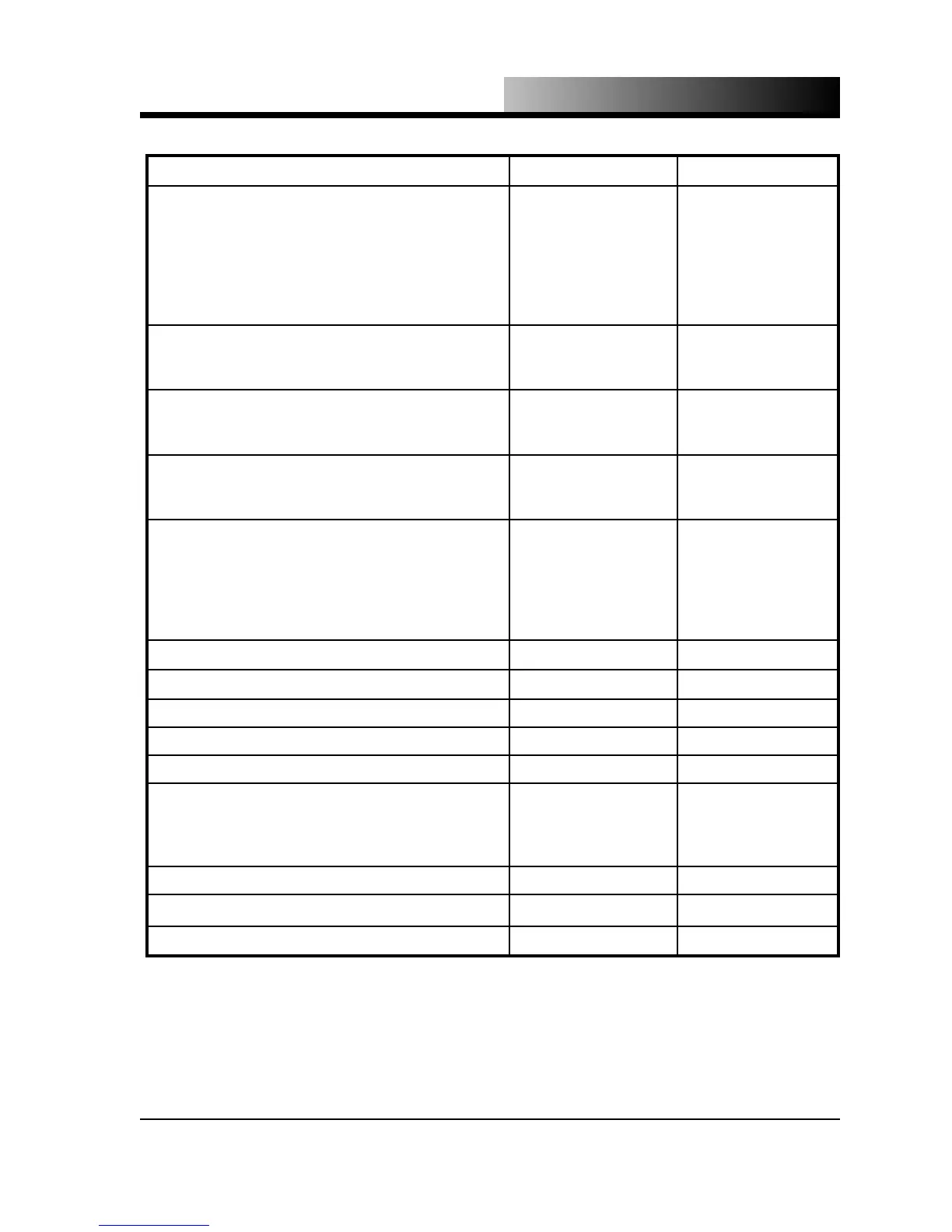
Molecular Weight 44.01 lb/lb-mol 44.01 kg/kg-mol
Vapor Pressure
at 70 °F (21.1 °C)
at 32 °F (0 °C)
at 2 °F (-16.7 °C)
at -20 °F (-28.9 °C)
at -69.9 °F (-56.6 °C)
at -109.3 °F (-78.5 °C)
838 psig
491 psig
302 psig
200 psig
60.4 psig
0 psig
57.79 bar
33.86 bar
20.83 bar
13.79 bar
4.17 bar
0 kPa
Density of the gas
at 70 °F (21.1 °C)
at 32 °F (0 °C)
0.1144 lb/ft
3
0.1234 lb/ft
3
1.833 kg/m
3
1.977 kg/m
3
Specific gravity of the gas
at 70 °F (21.1 °C)
at 32 °F (0 °C)
1.522
1.524
1.522
1.524
Specific volume of the gas
at 70 °F (21.1 °C)
at 32 °F (0 °C)
8.741 ft
3
/lb
8.104 ft
3
/lb
0.5457 m
3
/kg
0.5059 m
3
/kg
Density of liquid, saturated
at 70 °F (21.1 °C)
at 32 °F (0 °C)
at 2 °F (-16.7 °C)
at -20 °F (-28.9 °C)
at -69.9 °F (-56.6 °C)
47.6 lb/ft
3
58.0 lb/ft
3
63.3 lb/ft
3
66.8 lb/ft
3
73.5 lb/ft
3
764 kg/m
3
931 kg/m
3
1016 kg/m
3
1072 kg/m
3
1179 kg/m
3
Sublimation temperature (1 atm) -109.3 °F -78.5 °C
Critical temperature 87.9 °F 31.1 °C
Critical pressure 1070.6 psig 73.81 bar, abs
Critical density 29.2 lb/ft
3
468 kg/m
3
Triple point -69.9 °F at 60.4 psig -56.6 °C at 4.16 bar
Latent heat of vaporization
at 32 °F (0 °C)
at 2 °F (-16.7 °C)
at -20 °F (-28.9 °C)
100.8 Btu/lb
119.0 Btu/lb
129.6 Btu/lb
234.5 kJ/kg
276.8 kJ/kg
301.4 kJ/kg
Latent heat of fusion at -69.9 °F (-56.6 °C) 85.6 Btu/lb 199 kJ/kg
Density of liquid at 2 °F (-16.7 °C) 63.3 lb/ft
3
1015.9 kg/m
3
Latent heat of sublimation at -109.3 °F (-78.5 °C) 245.5 Btu/lb 571.0 kJ/kg
Table 1.1 - Physical Constants of Carbon Dioxide
 Loading...
Loading...