Page 46 of 46
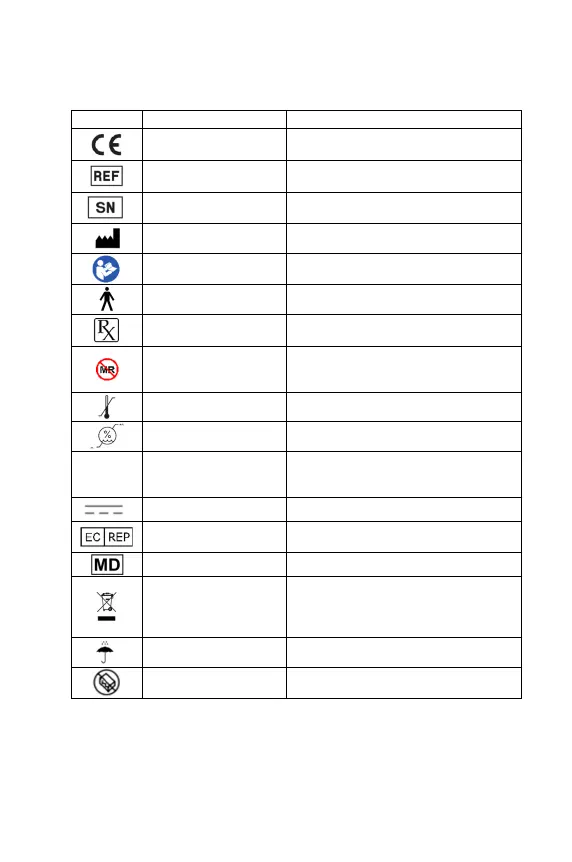
SYMBOL DEFINITIONS
Medical Devices Regulation
2017/745
CE Mark
ISO 15223-1:2016
Device Name
ISO 15223-1:2016
Serial Number
ISO 15223-1:2016 Manufacturer & Date of Manufacture
ES 60601-1:2005+A1:2012
Attention! See Instructions for Use
ES 60601-1:2005+A1:2012 Type B Applied Parts
N/A
CAUTION: Federal (USA) law restricts the sale
of this device by or on the order of a physician
ASTM F203
MRI Unsafe
Keep Away from Magnetic Resonance
Imaging (MRI) Equipment
ISO 15223-1:2021 Temperature Limits
ISO 15223-1:2021 Humidity Limits
IP21
ES 60601-1:2005+A1:2012
Solid Particle Protection Level 2 (>12.5mm);
Liquid Ingress Protection Level 1 (dripping
water)
ES 60601-1:20-05+A1:2012 Rated Input Power
ISO 15223-1:2016
Authorized Representative in the European
Community
ISO 15223-1:2021 Medical Device
IEC 60417
Waste of Electrical and Electronic Equipment
(WEEE) To indicate that separate collection
for waste electric and electronic equipment
(WEEE) is required
ISO 7000 Keep away from rain
ISO 15223-1:2021
Do not use if package is damaged and consult
instructions for use
 Loading...
Loading...