Page 2 of 46
for improving visual acuity through magnification to aid the process of cervical
intraepithelial neoplasia diagnosis, reducing false negative diagnoses, and have
been demonstrated in combination with telemedicine and image capture to allow
for remote diagnoses that are comparable to standard colposcopic examination,
thus expanding the ability for appropriately trained nurses to perform colposcopy
exams.
HOW SUPPLIED
The following components are
included with the MobileODT EVA
Pro:
● EVA Pro Digital Colposcope
Device
● Two Removable Lithium-Ion
Battery Packs
● Charging Base with A/C
Adapter and universal plugs
● Hard Shell Carrying Case
● Stand Connector
● Instructions for Use
● Optional Accessories:
Tripod / Wheeled Stand
CONTRAINDICATIONS
There are no contraindications associated with this device.
RESIDUAL RISKS
The EVA System should not touch the patient. There are minimal risks to the
patient. The system has been tested according to specific medical standards.
WARNING AND PRECAUTIONS
1. No modification of this equipment is allowed.
2. Local and/or other applicable laws may restrict the sale of this device to, or
to the order of, a physician or other proprietary licensed medical
professional. The EVA Pro may not be used unless you or your organization
meet the requirements and/or obligations under such laws and you assume
full responsibility for any illegal purchase and/or use of the device. The EVA
Pro should only be used by a trained and licensed operator, whose
accreditation is valid in the country and/or region in which the Procedures
are being conducted.
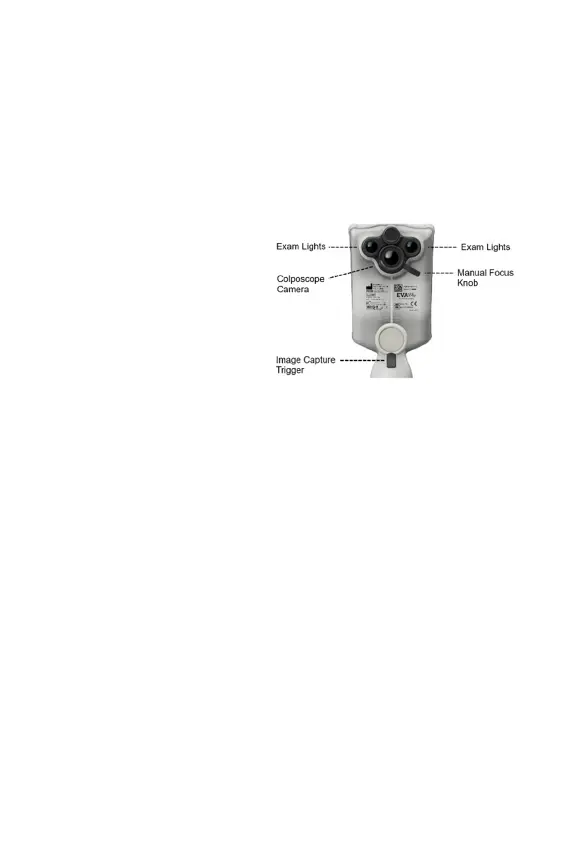
Figure 2: The EVA Pro device design
 Loading...
Loading...