©2022 Inogen, Inc. All rights reserved. Page 46 of 356 96-12100-01-01-A
15.2 PULSE VOLUME FLOW SETTINGS*
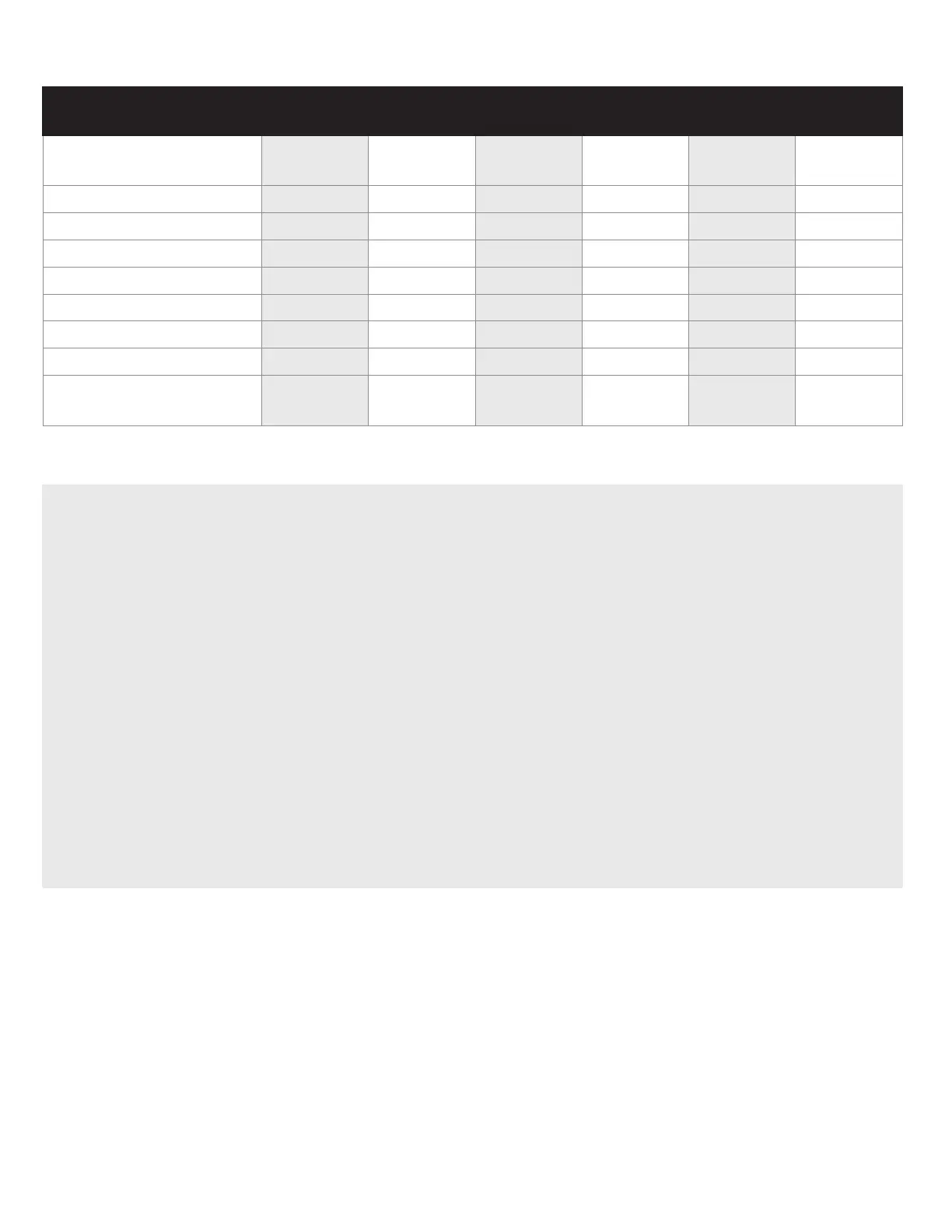
Inogen Rove 6 Pulse Volumes per Flow Setting
(mL/breath
± 15% per ISO 80601-2-67)
BREATHS PER
MINUTE
1 2 3 4 5 6
10
21.6 43.4 65.7 85.8 104.5 123.1
15
14.2 29.2 43.3 56.7 69.2 82.1
20
10.9 22.1 32.9 43.2 52.9 62.4
25
8.9 17.5 26.7 35.0 42.9 50.7
30
7.4 14.8 22.0 29.3 36.0 42.6
35
6.3 12.8 18.8 25.0 30.4 36.7
40
5.4 11.3 16.6 21.7 26.5 31.6
TOTAL VOLUME PER
MINUTE (ml/min)
210 420 630 840 1050 1260
15.3 ELECTROMAGNETIC COMPATIBILITY (EMC) INFORMATION
WARNING!
Risk of death, injury or damage
• Use of accessories, transducers and cables other than those specified or provided by the manufacturer of
this equipment could result in increased electromagnetic emissions or decreased electromagnetic immunity
of this equipment and result in improper operation.
• Avoid exposure to known sources of EMI (electromagnetic interference) such as diathermy, lithotripsy, elec-
trocautery, RFID (Radio Frequency Identification), and electromagnetic security systems such as anti-theft/
electronic article surveillance systems, metal detectors. Note that the presence of RFID devices may not be
obvious. If such interference is suspected, reposition the equipment, if possible, to maximize distances.
• Portable RF communications equipment (including peripherals such as antenna cables and external anten-
nas) should be used no closer than 30 cm (12 inches) to any part of the device, including cables specified by
the manufacturer. Otherwise, degradation of the performance of this equipment could result.
• The device should not be used adjacent to or stacked with other equipment. If adjacent or stacked use is
necessary, the device should be observed to verify normal operation. If operation is not normal, the device or
the other equipment should be moved.
Medical electrical equipment needs to be installed and used according to the EMC information in this manual.
This equipment has been tested and found to comply with EMC limits specified in IEC 60601-1-2. These limits are
designed to provide a reasonable protection against electromagnetic interference in a typical home environment.
This concentrator contains Transmitter Module IC: 2417C-BX31A. Contains FCC ID: N7NBX31A. This device com-
plies with Part 15 of the FCC rules. Operation is subject to the following two conditions: (1) this device may not
cause harmful interference, and (2) this device must accept any interference received, including interference that
may cause undesired operation.

 Loading...
Loading...