Instructions for use MULTIflex Coupling 465 RN – 0.553.1600, MULTIflex LUX Coupling 465 LRN – 0.553.1550
7 Reprocessing steps in accordance with ISO 17664 | 7.1 Preparations at the site of use
19 / 28
7 Reprocessing steps in accordance with ISO 17664
7.1 Preparations at the site of use
WARNING
Hazard from contaminated products.
Contaminated products are associated with an infection hazard.
4 Take suitable personal protective measures.
4 Reprocess the medical device right after treatment.
4 The medical device must be dry when transported to reprocessing.
4 To minimise the risk of infection during reprocessing, always wear protect-
ive gloves.
4 Remove straight and contra-angle handpieces from the medical device.
4 Remove all residual cement, composite or blood immediately.
4 Do not place in solutions or similar substances.
7.2 pre-cleaning
CAUTION
Never reprocess this medical device in an ultrasonic cleaner.
Malfunction and material damage.
4 Reprocess it in a washer disinfector only.
Requisite accessories:
▪ Tap water 30
o
C ± 5
o
C (86
o
F ± 10
o
F)
▪ Brush, e.g. medium-hard toothbrush
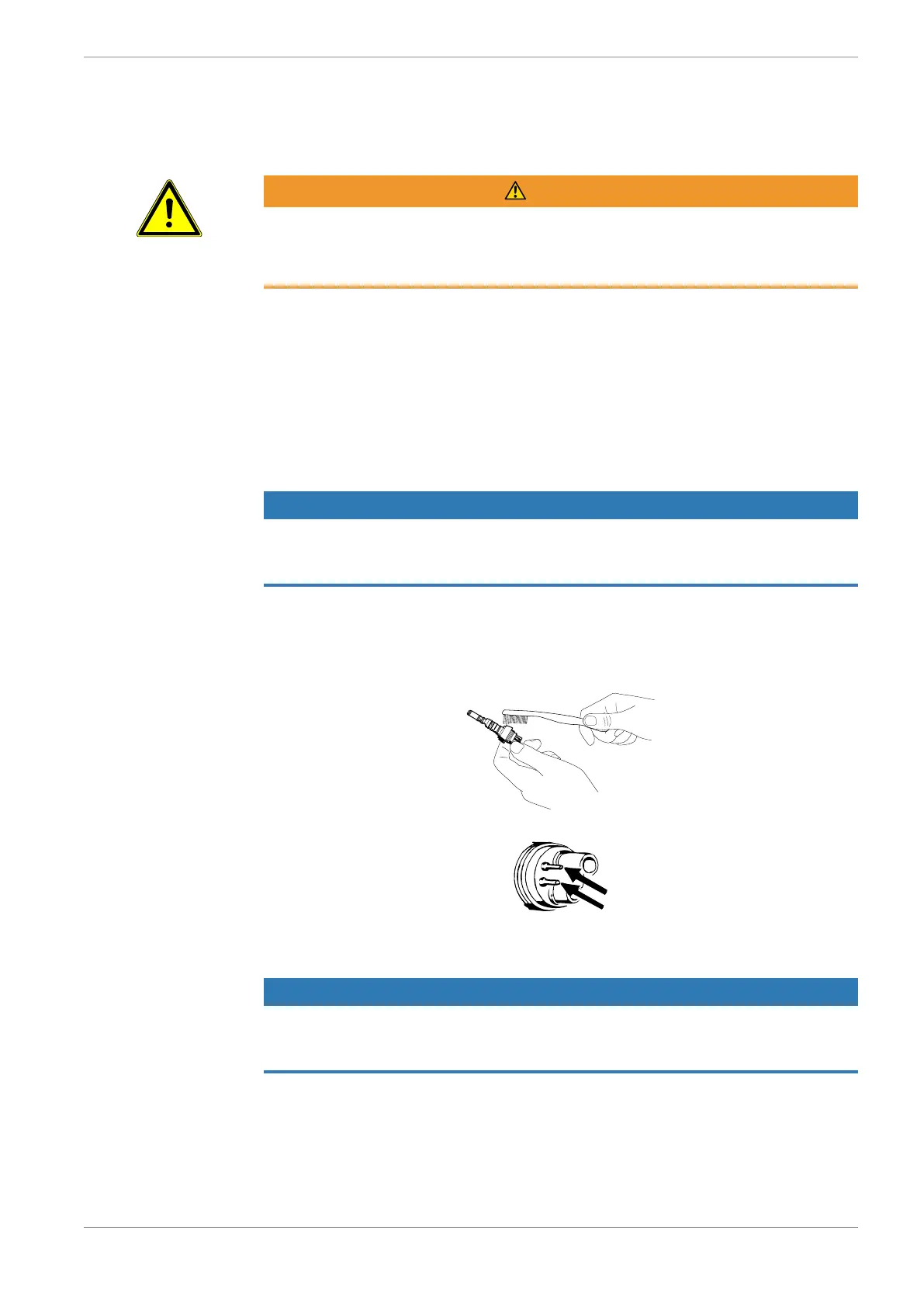
4 Brush off under flowing tap water.
4 Rinse the spray air and spray water tube with potable water.
7.3 Manual reprocessing
CAUTION
Never reprocess this medical device in an ultrasonic cleaner.
Malfunction and material damage.
4 Reprocess it in a washer disinfector only.
7.3.1 Manual internal and external cleaning
This product is not designed for manual internal and external cleaning.

 Loading...
Loading...