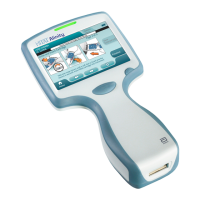
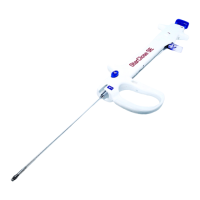
Symbol Descripon
Manufacturing facility
Axed in accordance with European Union Medical Device Regulaon 2017/745
and Radio Equipment direcve 2014/53/Annex II. Hereby, Abbo Medical declares
that this device is in compliance with the essenal requirements and other relevant
provisions of this regulaon and direcve.
The full text of the European Union Radio Equipment direcve 2014/53/EU
declaraon of conformity is available at the following internet address:
www.cardiovascular.abbo/euconformity.
This product operates in the 402‑405 MHz band with an eecve radiated power of
less than 25 µW ERP.
This product operates between 9 and 200 kHz with an H‑eld strength of less than
25 dBµA/m at 10 m.
The device contains a baery and the label is axed to this device in accordance
with European Council Direcve 2006/66/EC.
Return the device to
Abbo
Medical
when explanted or dispose as potenally
biohazardous material in accordance with medical pracce and applicable local,
state, and federal laws and regulaons.
53