1 General Information about the Device
1.5 Marking
1.5 Marking
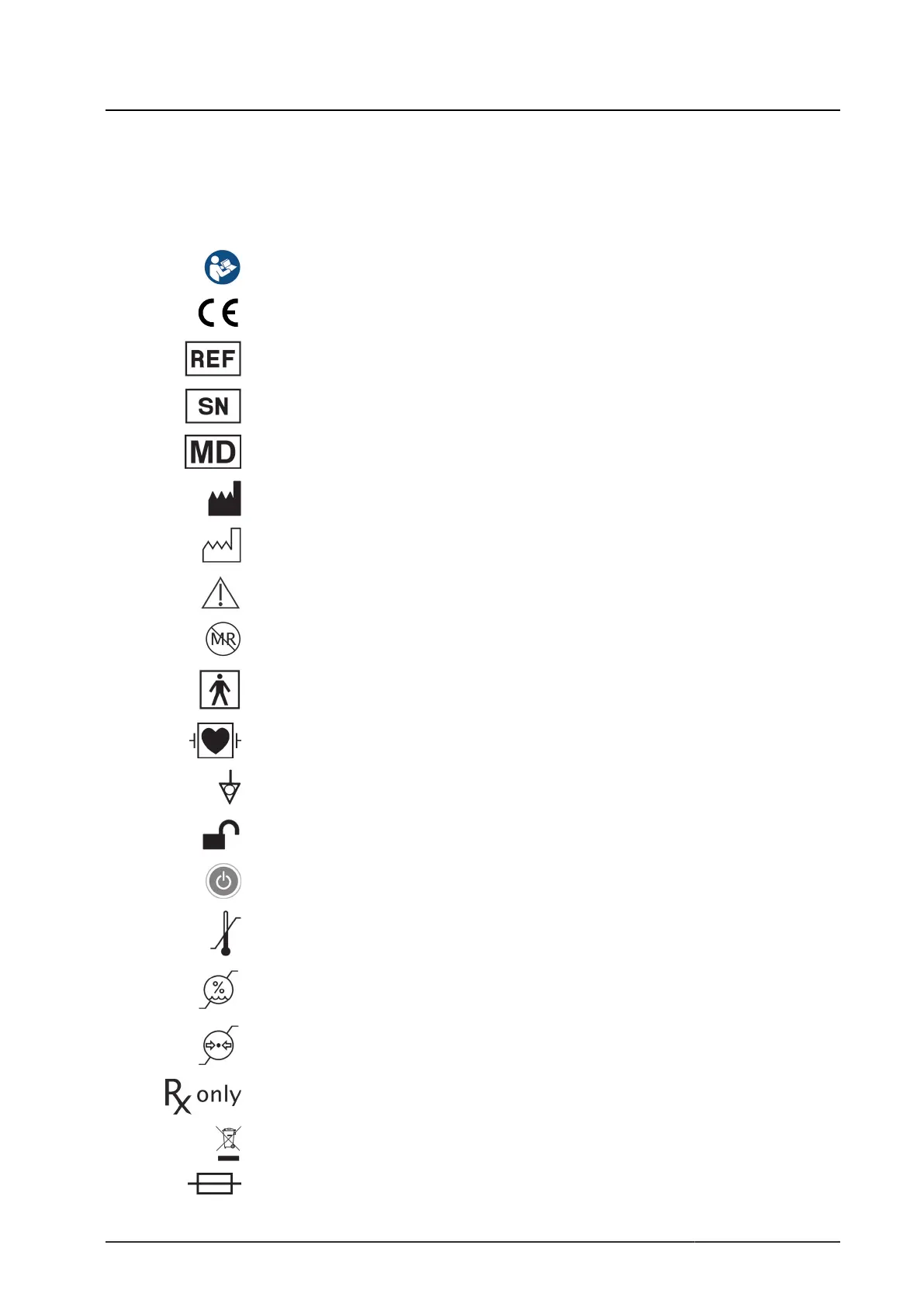
1.5.1 Pictograms and Information on the Device and Packaging
This section describes the different pictograms that feature on the product or
packaging.
Adhere to the instructions for use
CE marking
Item number
Serial number
Medical Device
Manufacturer
Date of manufacture
Caution (IEC 60601-1 3rd and 4th edition) /
Take note of accompanying documents (IEC 60601-1 3rd and 4th edition)
MR unsafe
Type BF applied part according to IEC 60601-1
Type CF defibrillation-proof applied part according to IEC 60601-1
Potential equalization
Release
ON / standby
Permissible storage and transport temperature
Permissible relative air humidity during storage and transport
Permissible atmospheric pressure during storage and transport
Caution: Federal (US) law restricts this device to sale by or on order of a physician
Separate collection for WEEE (waste of electrical and electronic equipment)
Fuse
en
Instructions For Use | 13 - Mar - 2020 | Version: B Page 49 of 78

 Loading...
Loading...