▪ Advise paents to not carry earbuds or headphones in their breast pocket or near implant area, and to▪
not allow earbuds or headphones to drape around the paent's neck so they hang on the chest. These
devices may contain a magnet or magnec material, or may emit RF signals that can interfere with the
pulse generator and may cause the pulse generator to pace abnormally.
Preparaon for Implantaon
Package Label. Before opening the sterile package, carefully read the label and verify that the package
contains the desired device. Do not implant the pulse generator if:
▪ The package is damaged or wet.
▪
▪ The dot on the ethylene oxide label is purple. Purple indicates that the package has not been sterilized.
▪
▪ The Use Before Date on the outer box and the tray has been exceeded. The Use Before Date reects the
▪
minimum baery voltage required to support the calculated baery longevity shown in the programmer's
on‑screen help.
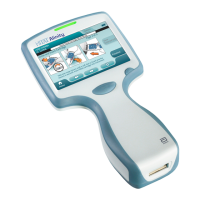
Verifying Operaon. To verify that the device is operang properly remove the magnet from the programmer
telemetry wand and establish communicaon:
▪ Inducve communicaon. Posion the
Merlin
™ PCS or
Merlin
™ 2 PCS telemetry wand over the package▪
and select Interrogate.
▪
RF communicaon. To establish RF communicaon between the device and the programmer and to
▪
troubleshoot communicaons problems, you must rst aach the Merlin antenna to the programmer.
Refer to the Merlin PCS or Merlin 2 PCS User's Manual that accompanies the programmer and the
Merlin antenna. Use the telemetry strength indicators to evaluate the communicaon.
If the device is RF‑compable, an icon in the upper le‑hand corner of the screen during the
programming session indicates the status of the RF communicaon link. If an RF icon does not appear on
21