TECHNICAL SPECIFICATIONS
49 OTOWAVE 302+ INSTRUCTION FOR USE
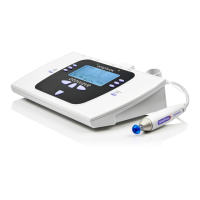
7. TECHNICAL SPECIFICATIONS
7.1. STANDARD AND REGULATORY
The CE mark indicates that Amplivox Ltd. Meets the requirements of Annex II of the Medical
Device Regulation 2017/745. TÜV Product Service, Identification No. 123, has approved the
quality system.
The Otowave 302+ Tympanometer is classified as a Class IIa device under Annex VIII of the EU
Medical Devices Regulation.
Standards and
Conformance
IEC 60601-1 (plus ES, CSA & EN deviations), Class II, Type B
applied part
IEC 60645-5, Type 2 Tympanometer
ANSI 3.39, Type 2 Tympanometer
256 x 64 pixels / 8 lines of 21 characters
270 x 70 x 175 mm / 10.63 x 2.75 x 6.89 inch
(excluding connections)
130 x 25 mm / 5.11 x 0.98 inch
Interconnection:
(probe to base)
1.5 m combined electrical cable and air tube
100-240 Vac; 50/60 Hz; 0.4 A
+15°C to +35°C / + 59°F to +95°F
30 % to 90 % RH (non-condensing)
Operating atmospheric
pressure:
Transport: storage
temperature:
-20°C to +70°C / -4°F to +94°F
Transport and storage
humidity:
10 % to 90 % RH (non-condensing)
Transport and storage
atmospheric pressure:
Time and date stamp applied to all recordings, and to the last
calibration date
English, German, French, Spanish, Portuguese, Italian
Any recording can be stored once the tympanogram is
viewed. Patient Initials (A-Z, 0-9, “-“) must be entered before
storage.
Patient Initials, Tympanogram and Reflex graphs and analysis
for Left Ear and/or Right Ear, Time and Date of recording,
which ears were tested, whether or not the record has been