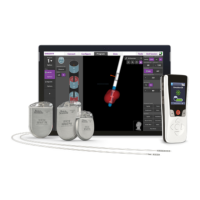
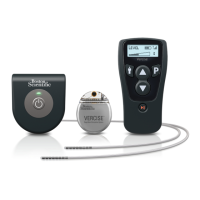
Vercise™ Deep Brain Stimulation Systems Information for Prescribers
92366226-02 iii of iv
Table of Contents
Device Description .......................................................................................... 1
Intended Use / Indications for Use .......................................................................... 2
Safety Information ........................................................................................... 3
Contraindications..................................................................................................... 3
Warnings ................................................................................................................. 4
Precautions ............................................................................................................. 7
Adverse Events ....................................................................................................... 13
Service and Maintenance................................................................................ 15
External Trial Stimulator (ETS) Maintenance .......................................................... 15
Cleaning the Charging System ................................................................................ 16
Cleaning the Remote Control .................................................................................. 16
Cleaning the Programming Wand ........................................................................... 16
Electromagnetic Compatibility ....................................................................... 17
EN 60601‑1‑2 Classication Information ................................................................. 17
Notice to the User Per Industry Canada Radio Standards Specications ............... 23
Essential Performance ............................................................................................ 24
Telemetry Information .............................................................................................. 24
Quality of Wireless Service ..................................................................................... 25
Troubleshooting Wireless Coexistence Issues ........................................................ 26
Wireless Security ..................................................................................................... 27
Rechargeable Stimulator Battery ................................................................... 28
Stimulator Battery .................................................................................................... 28
Recharge Estimate .................................................................................................. 28
Non‑Rechargeable Stimulator Battery........................................................... 29
Stimulator Battery .................................................................................................... 29
Elective Replacement .............................................................................................. 29
End of Service ......................................................................................................... 29
Limited Warranty ‑ IPGs .................................................................................. 30
Rechargeable Implantable Pulse Generator (IPG) .................................................. 30
Non‑Rechargeable Implantable Pulse Generator (IPG) .......................................... 31
Limited Warranty ‑ Leads ................................................................................ 32
Limited Warranty ‑ Externals .......................................................................... 33
Technical Support ........................................................................................... 34
 Loading...
Loading...