18
with the word “Ready”. The user can move to another PPR by scrolling through
the list by pressing either the up or down arrow in the central cluster. Once
finding the program of interest, the user selects the program by pressing the right
(Next) button.
3.1.4 Inside Flip Cover Label
Inside the flip cover is a list of all the PPR’s available in the device. The software
refers to the PPR’s by number. The flip cover label seen in Figure 5 relates the
PPR # to a typical clinical application.
Fig 5. PPR List
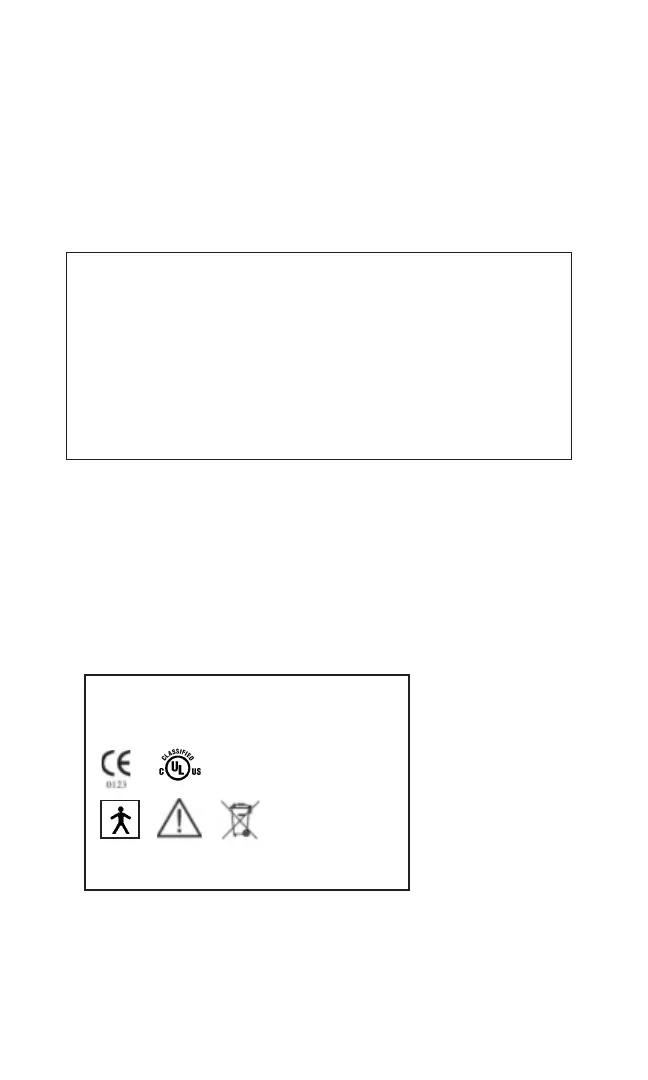
3.1.5 Backside Labeling
The labeling found on the back of the device, Figure 6, details the standards to
which this device has been tested, CE Mark and UL approvals. Federal law (USA)
restricts this device to sale by or on the order of a physician. Additionally, the
user is explicitly warned to not use the device for transthoracic stimulation.
Fig 6. Regulatory Labeling
NMES PPRs
PPR TYPE APPLICATION
1 NMES LG Muscle Re-education
2 NMES SM Muscle Re-education
3 NMES ROM/Atrophy
4 NMES ROM/Atrophy
5 NMES Gait
6 NMES Spasm
7 TENS Pain
Custom USER 1
Custom USER 2
High Volt PPRs
PPR TYPE APPLICATION (CH1 ONLY)
1 HV(-) Increase Circulation
(Red Active/treatment wire is
Negative)
2 HV(+) Increase Circulation
(Red Active/treatment wire is
Positive)
Custom USER 1
Custom USER 2
NEUROMUSCULAR
STIMULATOR
Empi, St. Paul, MN 55126 USA
NOT FOR TRANSTHORACIC STIMULATION
CAUTION: Federal law (USA) restricts this device to sale by or on the
order of a physician.
UL 2601-1, CAN/CSA C22.2
No. 601.1-M90
11N1
00962.indd 1800962.indd 18 1/23/07 9:23:02 PM1/23/07 9:23:02 PM
 Loading...
Loading...