Roche Diagnostics April 2007
Service manual · Version 2.0 11
91xx pH/Electrolyte Analyzers
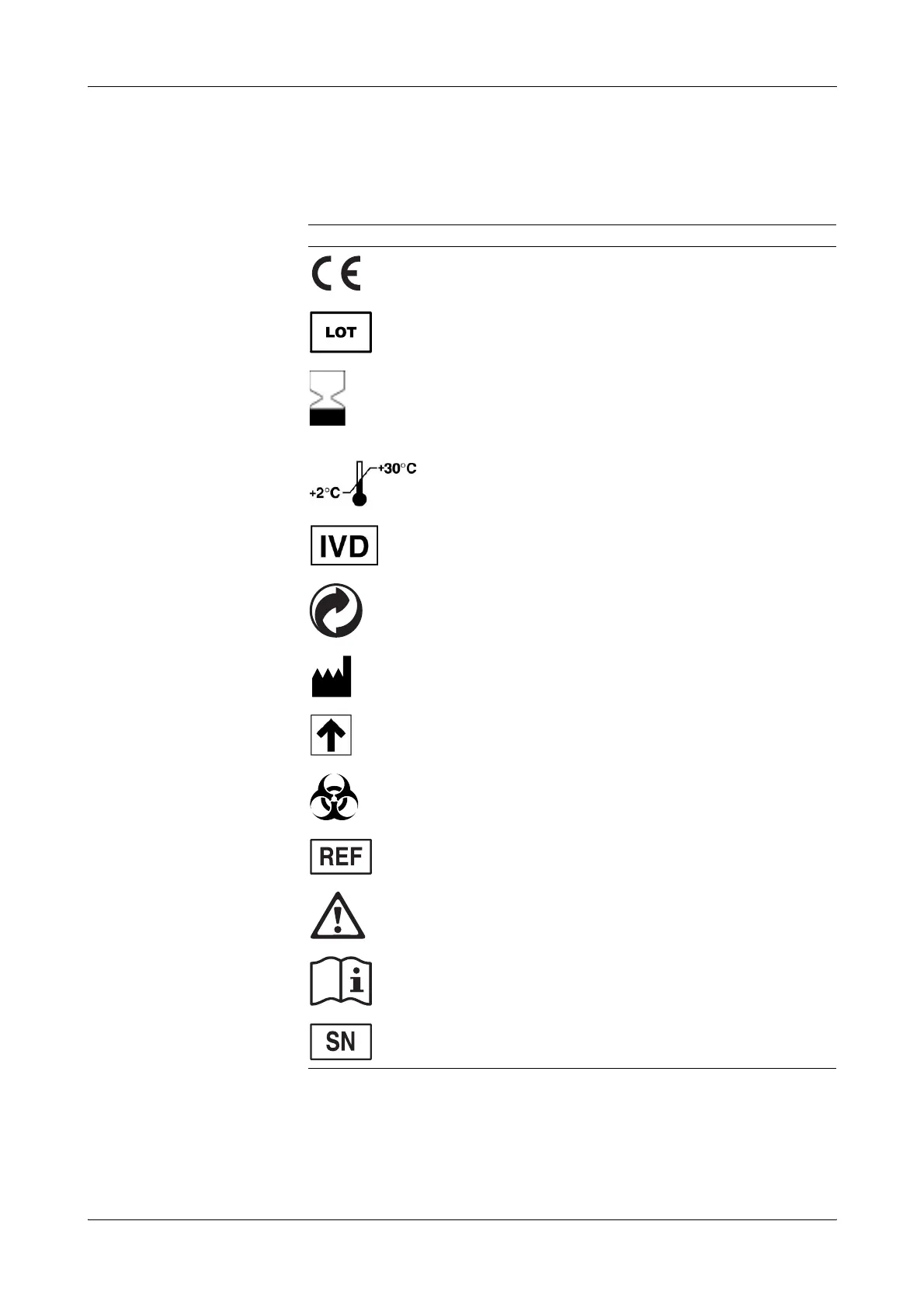
IVD symbols Instruments: applicable for 9180 Electrolyte Analyzers SN > 12001.
The IVD symbols are used in accordance with DIN EN 980:2003, ISO 15223:2000 (as
per the In Vitro Diagnostics Directive 98/79/EC), and DIN EN ISO 780:1997.
Symbol Description
This product complies with the requirements in the directive
98/79/EC on in vitro diagnostic medical devices.
Lot designation
Consumables: use by... (expiry date)
The consumables must be completely consumed by the indicated
date.
If a day is not indicated, apply the last day of the respective month.
Store at ...
The conditions necessary to preserve the product's shelf life before
opening.
For in vitro diagnostic use
"Grüner Punkt" (in Germany)
Manufacturer — according to directive 98/79/EC on in vitro
diagnostic medical devices
Store upright
Risk of infection!
(according to the standard DIN ISO 15223-1:2005) (Consumables)
Catalogue number
Caution (refer to accompanying documents). Please refer to safety-
related notes in the manual, accompanying this instrument.
Please consult instructions for use
Serial number (model plate)
 Loading...
Loading...