Chapter 7 CLEANING AND DISINFECTION
All information in this guide applies to ST, U/S, and EX modalities, unless otherwise noted.
Introduction
This chapter includes detailed cleaning and disinfection instructions for the Mammotome revolve Dual Vacuum-
Assisted Biopsy System per ISO 17664.
Users in North America should also refer to appropriate sections of AORN Standards & Recommended Practices
for additional guidance on cleaning and disinfection. All other localities should refer to appropriate guidelines.
The cleaning agents and disinfectants specified for use in these instructions have been validated for use with the
Mammotome revolve Biopsy System. The use of cleaning agents or disinfectants other than those specified in
these instructions should be assessed for equivalency before using. Technical data sheets are typically available
through the cleaning/disinfection agent manufacturer’s web pages to assist in this assessment. Any cleaning or
disinfection process, including tools and solutions, may influence the wear of a device or equipment.
There are no limitations for time of storage of any disinfected device prior to use and no additional support
systems are required.
PRECAUTION: Before cleaning or disinfecting the system, turn off the system and disconnect the power cord
from the power source. No further disassembly required.
NOTE: After cleaning and disinfecting the system, refer to the “Preventative Maintenance” section for
maintenance information. Inspect the devices for non-cosmetic damage prior to use, dispose or return
device to the manufacturer if damage is found.
WARNING: Do not attempt to sterilize the holsters, control module, cart, remote keypad, or remote footswitch
through autoclave, ethylene oxide, radiation, or plasma sterilization procedures; do not process the
devices through an automated washer-disinfect or ultrasound bath; do not spray the devices with
fluids or submerge in fluids; this may damage the instrument. If the devices are cleaned improperly,
the warranties may be void. Do not transport the device in a container prior to cleaning.
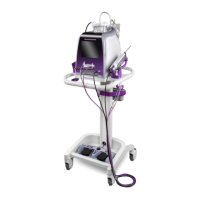
Cleaning Instructions for the Control Module, Cart, and Remote Footswitch
Cleaning the Control Module, Cart, and Remote Footswitch: Option 1
After each use, manually clean the control module, cart, and remote footswitch thoroughly utilizing a pH-
neutral enzymatic detergent. Proceed with manual cleaning utilizing the following procedure:
1. Prepare a pH-neutral detergent or pH-neutral enzymatic detergent according to the manufacturer’s
directions.
2. Use a soft, clean cloth lightly moistened with the cleaning solution to manually clean all surfaces,
including crevices.
3. To rinse, wipe thoroughly using a soft, clean cloth lightly moistened with lukewarm (27°C–44°C) tap
water.
4. Dry with a clean, soft cloth. Do not expose the device to heat greater than 54°C.
5. Inspect the device for visible contamination. If contamination is present, repeat steps 1-5 until no
visible contamination is present.
 Loading...
Loading...