V 1.31
2021. 05 166 / 171 Doc. # : BN-OP-C7
Guidance and manufacturer’s declaration – electromagnetic immunity
The Cardio7e/Cardio7 is intended for use in the electromagnetic environment specified below.
The customer or the user of the should assure that it is used in such an environment.
Electromagnetic environment -
guidance
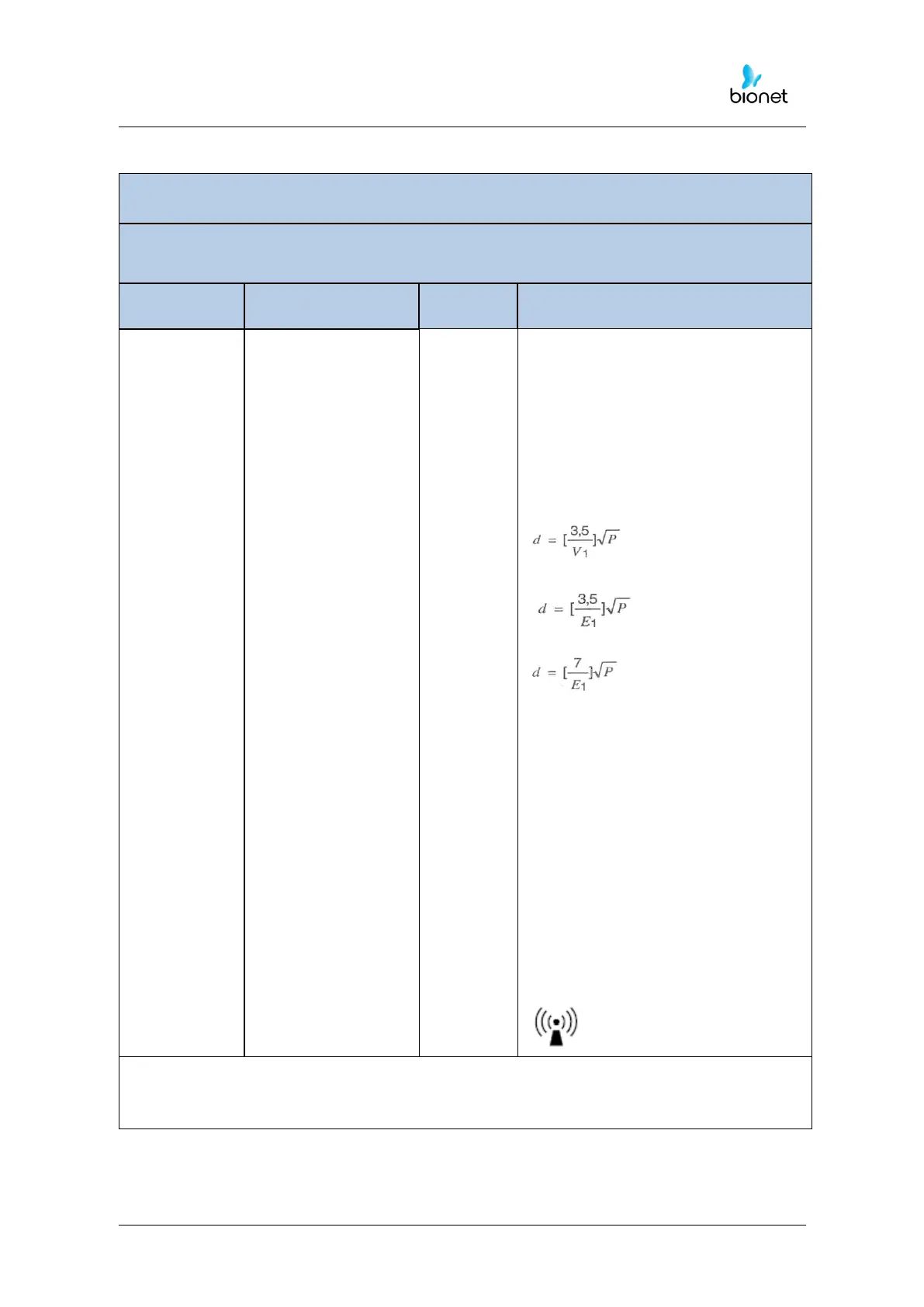
Conducted RF
IEC 61000-4-6
Portable and mobile RF
communications equipment should be
used no closer to any part of the
Cardio7e/Cardio7, including cables,
than the recommended separation
distance calculated from the equation
applicable to the frequency of the
transmitter.
Recommended separation distance
Radiated RF
IEC 61000-4-3
80 MHz to 800 MHz
800 MHz to 2,5 GHz
where P is the maximum output power
rating of the transmitter in watts (W)
according to the transmitter
manufacturer and d is the
recommended separation distance in
meters (m).
Field strengths from fixed RF
transmitters, as determined by an
electromagnetic site survey,
a
should be
less than the compliance level in each
frequency range.
b
Interference may occur in the vicinity of
equipment marked with the following
symbol:
NOTE 1 At 80 MHz and 800 MHz, the higher frequency range applies.
NOTE 2 These guidelines may not apply in all situations. Electromagnetic propagation is affected
by absorption and reflection from structures, objects and people.
 Loading...
Loading...