40
P073GUI001 - e-Celsius Medical Connect System User Manual.4 - EN
In this chapter you will nd the information necessary to ensure that your medical device is installed and
put into operation under the best possible conditions in terms of electromagnetic compatibility. The indi-
vidual cords of the medical device must be kept away from each other. Some types of mobile telecommu-
nication devices such as cell phones may interfere with the medical device. The recommended separation
distances in this chapter must therefore be observed.
The medical device must not be used close to another device or placed on top of it. If this cannot be
avoided, it is necessary to check its proper functioning under the conditions of use before use. The use of
accessories other than those specied or sold by BodyCAP as replacement parts may result in increased
emission or decreased immunity of the medical device.
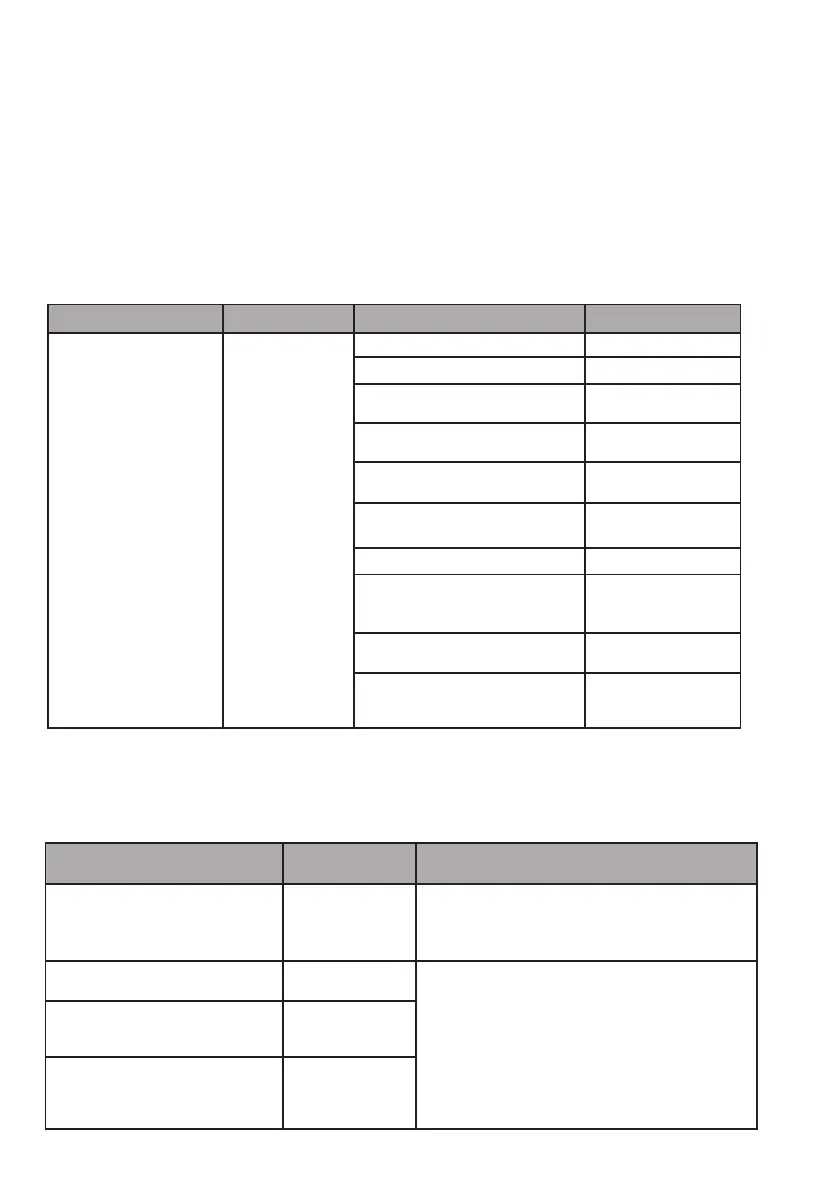
Cable length
Cables and accessories Maximum length Type of test In compliance with:
Cables/Cords < 3m
RF emission CISPR 11, Classe B
Emission of harmonic currents IEC 61000-3-2
Fluctuation and ickering of the
voltage
IEC 61000-3-3
Immunity to electrostatic discharges IEC 61000-4-2
Radiated immunity - Electromagnetic
elds
IEC 61000-4-3
Immunity to fast electrical transients
in bursts
IEC 61000-4-4
Shock wave immunity IEC 61000-4-5
Shock wave immunity Driving
immunity - Disturbance of the radio-
frequency line
IEC 61000-4-6
Radiated immunity - Magnetic elds IEC 61000-4-8
Immunity to voltage dips, short inter-
ruptions and voltage variations
IEC 61000-4-11
Guidelines and manufacturer's declaration of conformity regarding electromagnetic emissions
The e-Celsius® Medical System is intended for use in the electromagnetic environment described in the
table below. The user and installer should therefore ensure that the medical device is used in the environ-
ment described below.
Test Emission Compliance Electromagnetic environment Remarks
Disruption of radiation
electromagnetic (Radiated Emissions)
(CISPR11)
Group 1
The medical device uses RF energy for its
internal functioning
RF emission
(CISPR11)
Class B
The medical device is suitable for use in a health care
facility environment professional.
Current emission
harmonics
(IEC 61000-3-2)
Class A
Voltage variations, voltage uctua-
tions and icker
(IEC 61000-3-3)
Compliant
 Loading...
Loading...