9
P073GUI001 - e-Celsius Medical Connect System User Manual.4 - EN
Cleaning
The capsule is delivered sterile (ethylene oxide) in an individual packaging. It is not designed to
be cleaned with hydroalcoholic solutions.
The e-Celsius capsule® Medical must not be autoclaved under any circumstances, otherwise
it will be permanently damaged.
The other elements of the system (Watch & Activator) can be cleaned with a hydro-alcoholic
gel wipe.
European regulation REACH 1907/2006/EC
In response to the requirement of Article 33.1 of the REACH Regulation, we inform users of the
presence of the SVHC substance «Octyl Tin Stabiliser» in a concentration greater than 0.1%
mass / mass in the capsules. This substance is included in the candidate list published on June
15, 2015 under the CAS number 15571-58-1
(http://echa.europa.eu/fr/candidate-list-table).
The e-Celsius® Medical Connect System must be used in hospitals, clinics and exclu-
sively by health care personnel (doctors, nurses...). This includes HAD, EHPAD, home
health...
- It is also recommended to avoid splashing water on the blister pack and
exposure to sunlight. Storage at lower or higher temperatures may affect the operating
time and performance of the system.
- The shelf life of the e-Celsius® Medical capsule is indicated by an expiration
date on the blister pack. After this date, the sterility, performance and autonomy of the
device are no longer guaranteed.
- The device is designed to operate in an altitude range of 0 to 2000m.
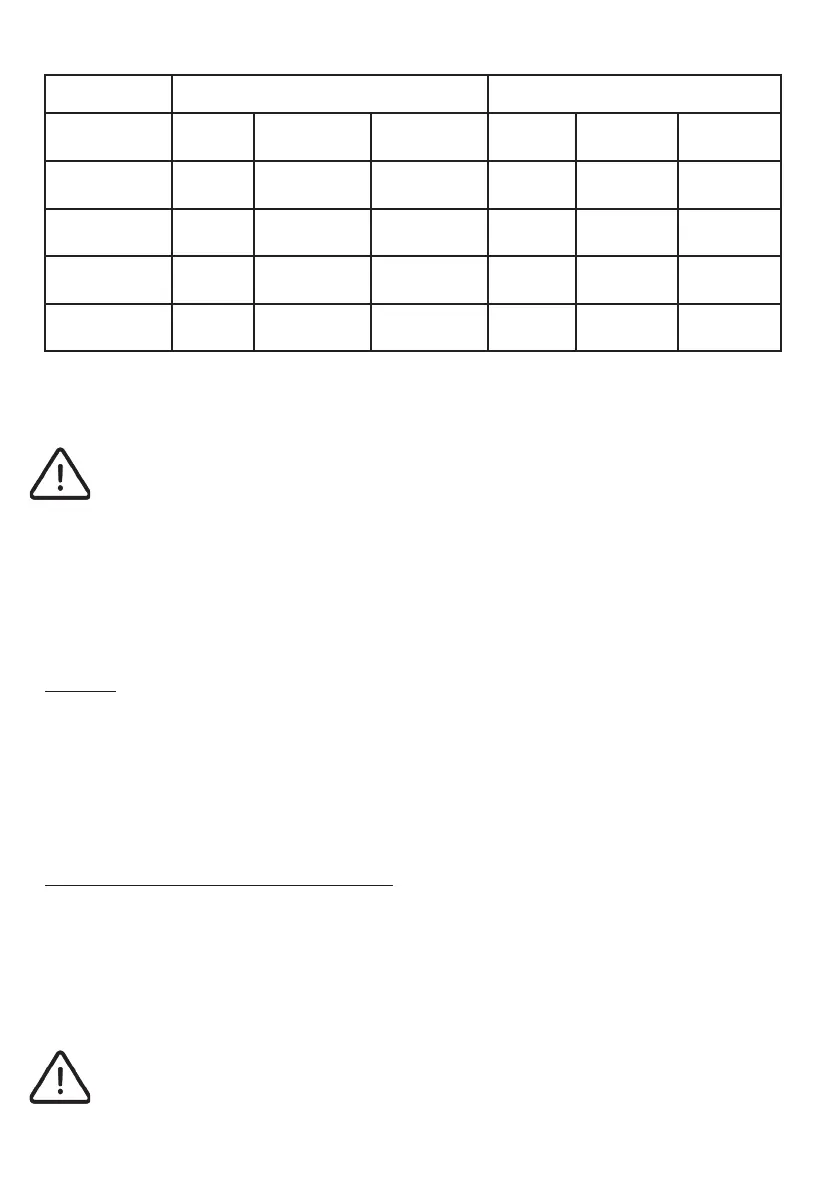
Storage/Transport Use
Temp °C Humidity
%
Pressure HPa Temp °C Humidity
%
Pressure HPa
P022 Capsules
5-35°C 30%-80% 700-1060hPa 25-45°C 100 700-1060hPa
P110 Watch
5-35°C 30%-80% 700-1060hPa 5-40°C 15% - 90% 700-1060hPa
P030 Activator
0-45°C 30%-80% 700-1060hPa 5-40°C 30%-80% 700-1060hPa
P073 System
5-35°C 30%-80% 700-1060hPa 5-40°C 30%-80% 700-1060hPa
Table 1: Environmental conditions
 Loading...
Loading...