12
COMPACT PIEZO LED
CAUTION: In the case that the end
user, when operating in his or her own
medical study or clinic, must subject the
electro-medical equipment and systems to
periodical inspections in order to adhere to
imposed requirements, the test procedures
that must be applied to electro-medical
equipment and systems to evaluate safety
must be performed in line with norm EN
62353‘Electro-medicaldevices-Periodical
inspections and tests to be performed after
repair interventions on electro-medical
devices’.Theintervalforperiodicchecks,in
the intended operating conditions is one year
or 2000 hours of use, depending on which of
thesetwoconditionsoccursrst.
WARNING: Riskofexplosion.
The device cannot operate in environments
where there are saturated atmospheres of
ammablegases(aestheticmixtures,oxygen,
etc.).
CAUTION: Theelectricalcontactsinside
thecordandhandpiececonnectorsmust
be dry. Before connecting the handpiece to
its cord, check that the electrical contacts are
perfectly dry, on both parts. If necessary dry
them with compressed air.
WARNING:OnlyuseoriginalMectron
inserttips,accessories,andspareparts.
CAUTION: It is prohibited to make any
changes to the device.
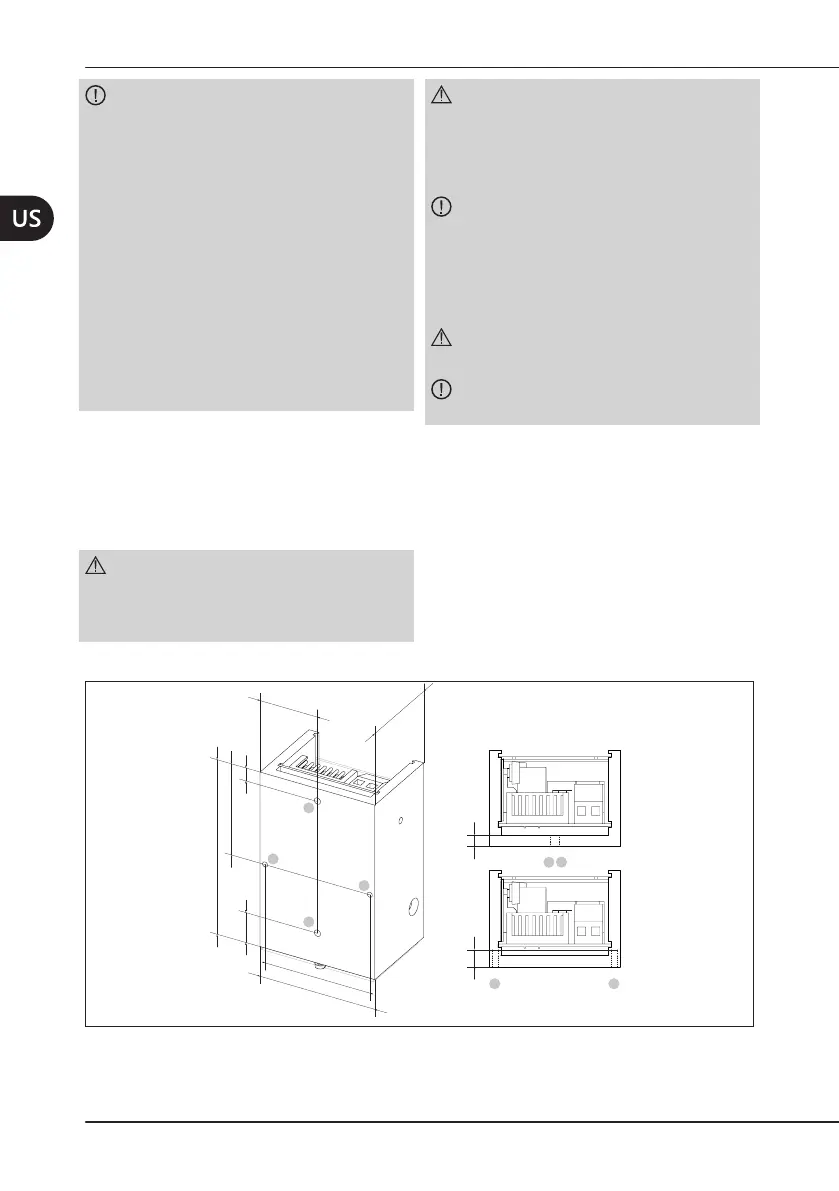
4.2 MechanicalInstallation
Figure2apagina12 shows the dimensions of the module.
The module can be secured to the dental unit by means of two M3 threaded holes (Ref. A and
B),oralternatively,bymeansoftwoholeswithdiameter2mm(Ref.CandD).
WARNING: The M3 screw used to secure
the module must not penetrate the inside of
said module for more than 4mm as it may
damage the circuits.
The holes with diameter 2 mm have a
maximumdepthof6mm.
The module is also supplied with holes on the
sides, but these must NOT be used to secure
it. The holes on the sides are used to attach
several internal components.
5,0012,00
70,00
36,50
46,00
2,00
2,00
42,00
M3
M3
23,00
34,00
A
A
B
B
C
C
D
D
MAX
4,00
M3
MAX
6,00
2,00 2,00
Figure2 – Dimensions of the module.

 Loading...
Loading...