APPENDIX A - DICOM Statement
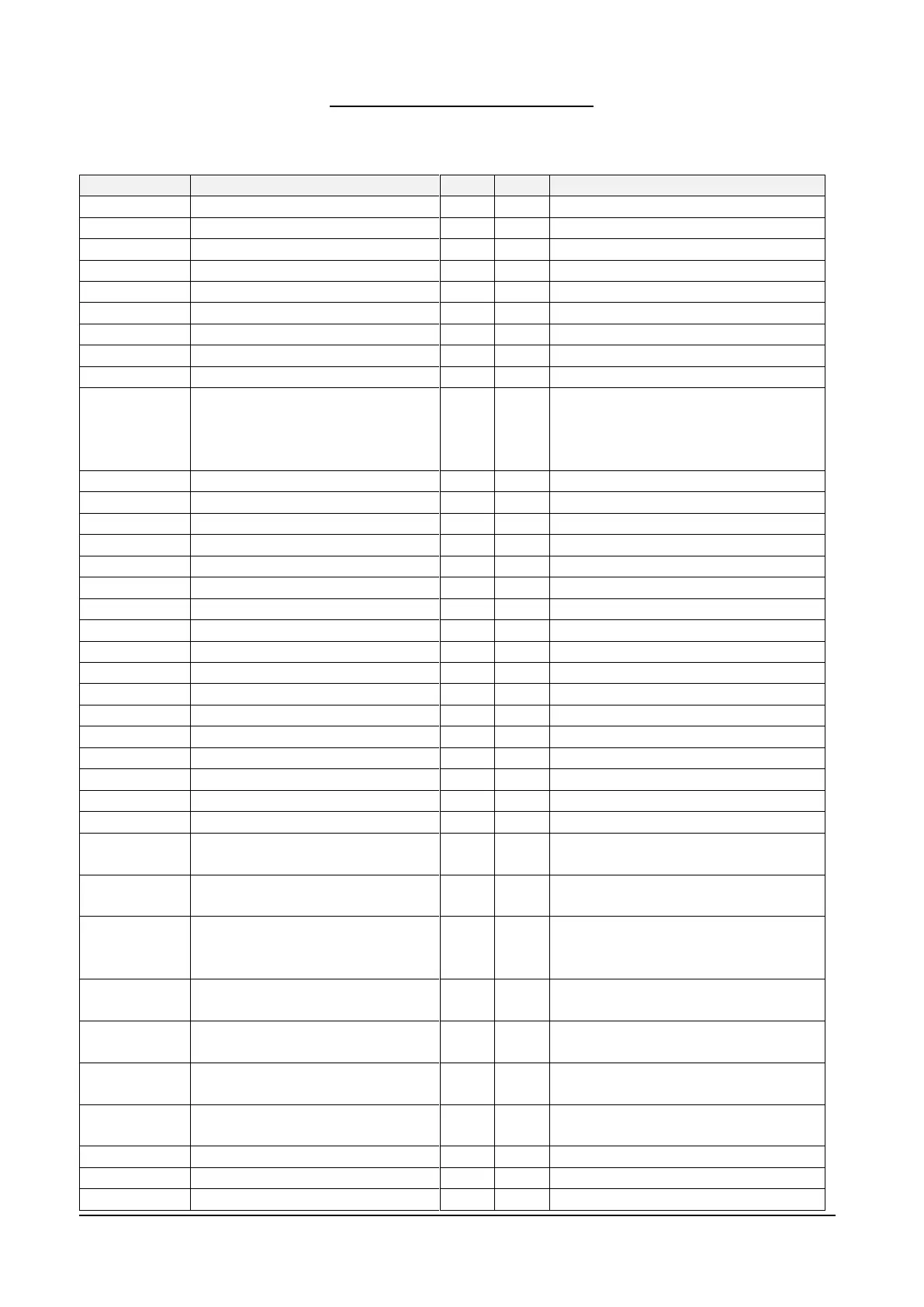
DRS can export a DICOM-compliant file format. Refer to the table below for the list of supported
tags.
File Meta Information Version
Media Storage SOP Class UID
1.2.840.10008.5.1.4.1.1.77.1.5.2
Media Storage SOP Instance UID
1.2.826.0.1.3680043.8.641
Implementation Version Name
Source Application Entity Title
1.2.840.10008.5.1.4.1.1.77.1.5.2
CenterVue ANSI registration code
(unique id number) plus machine id
and actual exam id.
Example: 2.16.840.1.114496.6.4
Referring Physician's Name
Value entered in Settings | Exam |
Advanced | Study Description
Value entered in Settings | Exam |
Advanced | Series Description
Value entered in the patient’s Last
Name and First Name fields (last +
first)
Value entered in the patient’s “code”
field
Value entered in the patient’s date of
birth fields
Value entered in the patient’s gender
field
Value entered in the patient’s “code”
field
 Loading...
Loading...