Care instructions KaVo uniQa
2 Reprocessing steps in accordance with ISO 17664 | 2.7 Reprocessing the three-function and multifunctional
handpiece
21 / 56
2.7.1 Preparations at the site of use
WARNING
Hazard from contaminated products.
Contaminated products are associated with an infection hazard.
4 Take suitable personal protective measures.
CAUTION
Danger of injury if the valve body is hot (heating elements, high-pres-
sure lamp).
Risk of burn injury.
4 Switch off the unit at the main switch.
4 Allow the handpiece to cool down after long use.
4 Remove gross soiling immediately.
4 Reprocess the medical device right after treatment.
4 Leave the gripping sleeve and cannula on the instrument, and activate the
air and water for 60 seconds.
4 The medical device must be dry when transported to reprocessing.
4 To minimise the risk of infection during reprocessing, always wear personal
protective equipment.
4 Do not place in solutions or similar substances.
2.7.2 Disassembly
WARNING
Incomplete reprocessing.
Infection hazard.
4 To ensure complete reprocessing of all parts, the medical device needs to be
disassembled before reprocessing.
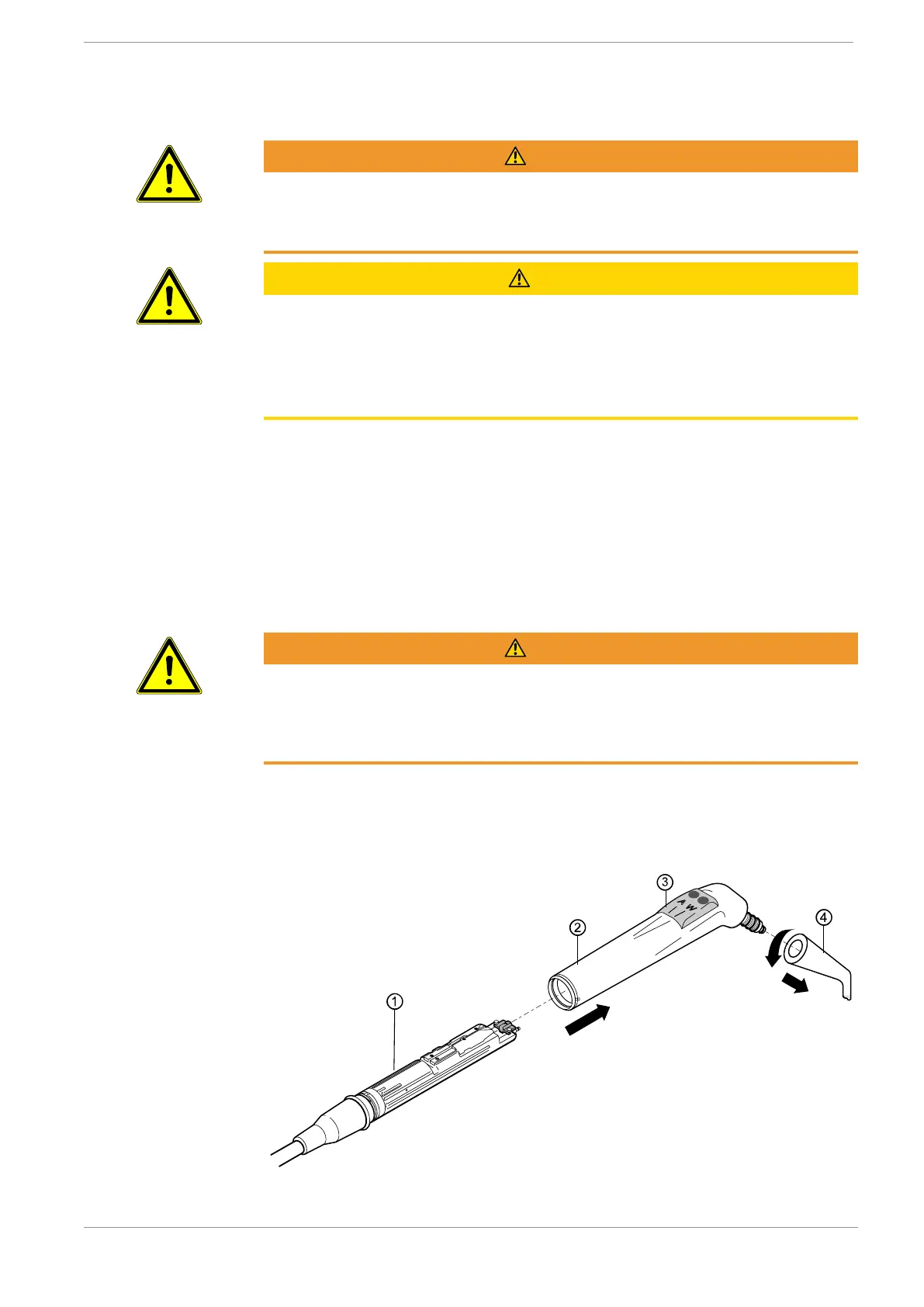
4 Hold the three-function or multifunctional handpiece by the gripping sleeve
② and take off the cannula ④ with a slight twisting motion.
4 Pull the gripping sleeve ② off the valve body ①.
4 Detach the key part ③ from the gripping sleeve ②.
 Loading...
Loading...