85
Annex B
Customer Returns Notification Form
This form must be stuck to the outside packaging of the returning
instrument and emailed to technicalsupport@bwbtech.com prior to
the instruments return. Please contact BWB on +44 (0) 1787 274
870 or speak to your local distributor should you require any advice
in completing this form. An A4 print ready version can be found on
your memory stick supplied with the instrument or alternatively
downloaded from the BWB Customer Online Portal.
Your order may be delayed if this form is not fully filled in,
please use block capitals and the English language.
PO or RMA number:
BWB reference number:
Unit Serial Number:
Instrument fault / description of return:
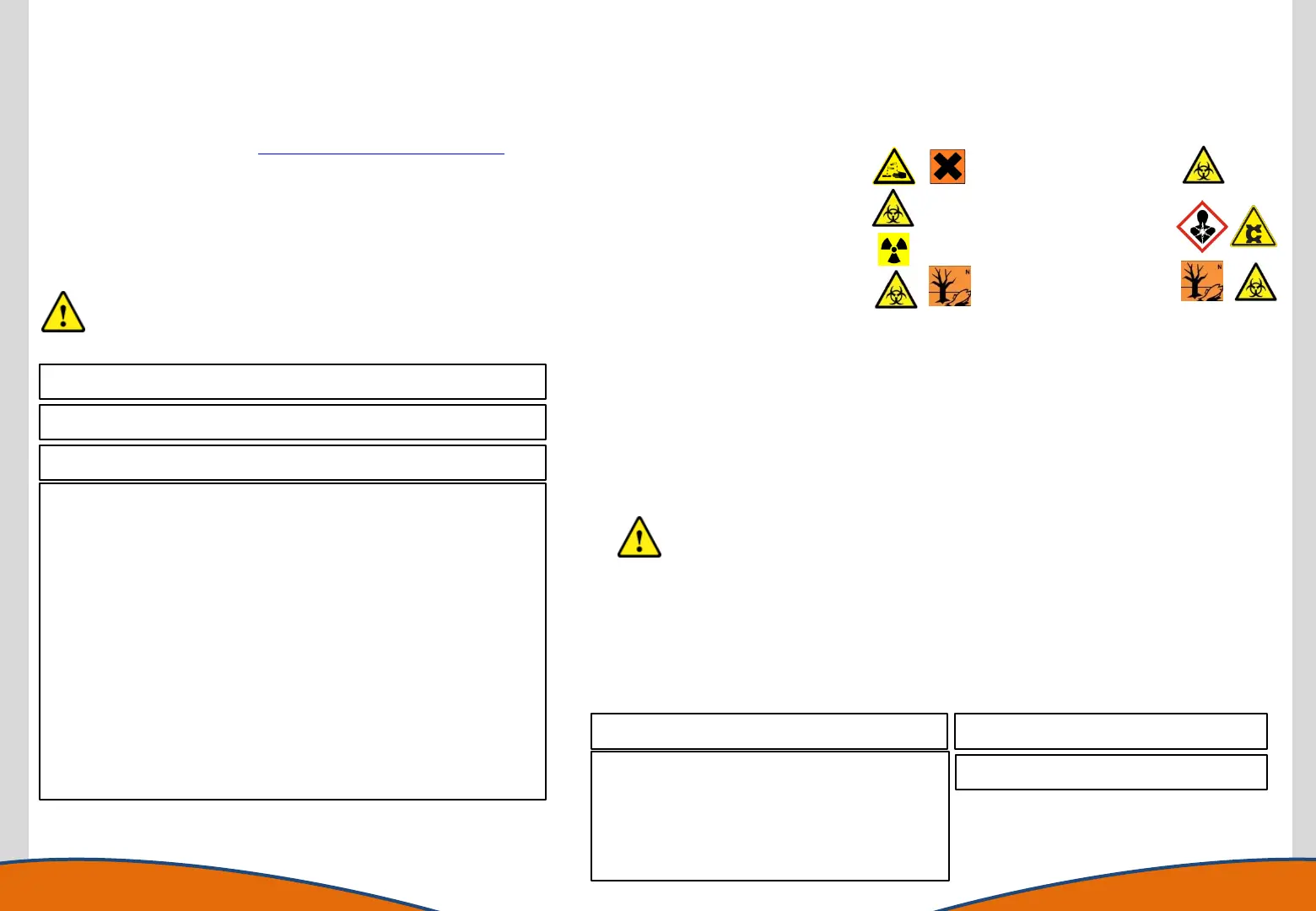
Contamination Information: Please tick all that apply. (The warning symbol may not
dictate actual contaminant).
[ ]Acids (corrosive and non-corrosive)
[ ]Blood (or products of)
[ ]Urine (or other human liquids)
[ ]Cancer-Causing
(carcinogenic)
[ ]Radioactive
[ ]Organic Solvents
[ ]Plant / Biological substances
[ ]Instrument not used
[ ]Other___________________
Unless otherwise specified by BWB Technologies UK , the “wet assembly”
(nebuliser, mixing chamber, burner and drain cup) are to be thoroughly
washed with an agent to rid them of relevant contamination. The outside of
the instrument is to be washed at minimum with BWB recommended
cleaning agent. If there is a cause to suspect the chassis of the instrument
has faced contamination it must be washed with an agent to remove it.
Material Safety Data Sheets must be attached for any media that has
contaminated the instrument.
I certify that the information provided is true and accurate. I understand
that I may be held liable for any contamination related injury or health
hazard caused to personnel during the handling and testing of this
instrument. I understand that the instrument will be returned to me at my
expense if this information is not fully complete or the instrument is clearly
contaminated.
Name: Date:
Signature:
Position:
 Loading...
Loading...