228
Eversense E3 CGM User Guide
20
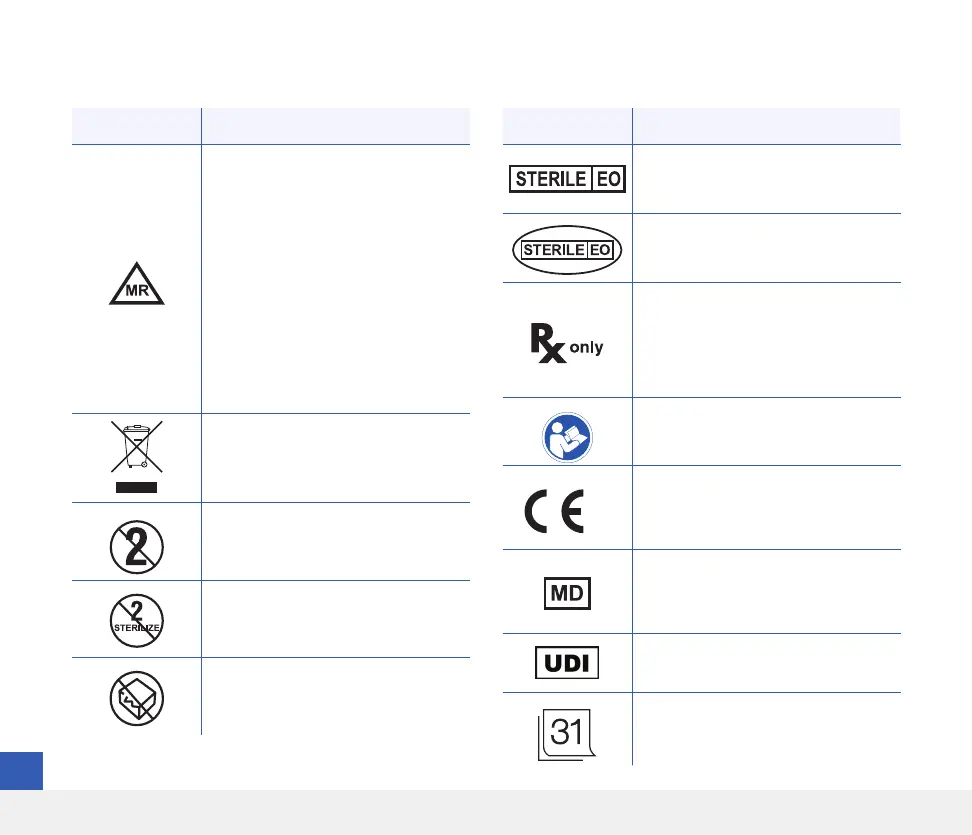
Symbol Explanation
MR Conditional. An item with
demonstrated safety in the MR
environment within defined
conditions including conditions
for the static magnetic field, the
time-varying gradient magnetic
fields and the radio frequency
fields. Please refer to the MRI
Safety Information section for
complete information.
European Union WEEE Directive
2012/19/EU
Single use only
Do not re-sterilise
Do not use if package is damaged
Symbols on Packaging and Devices (continued)
Symbol Explanation
Sterilised using Ethylene Oxide
Single sterile barrier: Sterilised
using Ethylene Oxide
U.S. (Federal) law restricts the sale
of the Eversense E3 CGM System to
sale by or on the order of a doctor
Follow instructions for use
2797
Marking certifies that the device
meets Medical Device Regulation
2017/745
Medical Device (Note: On the
implant card, this indicates the
device name)
Unique Device Identifier
Date of insertion
 Loading...
Loading...