36
10.4 Hygiene plan
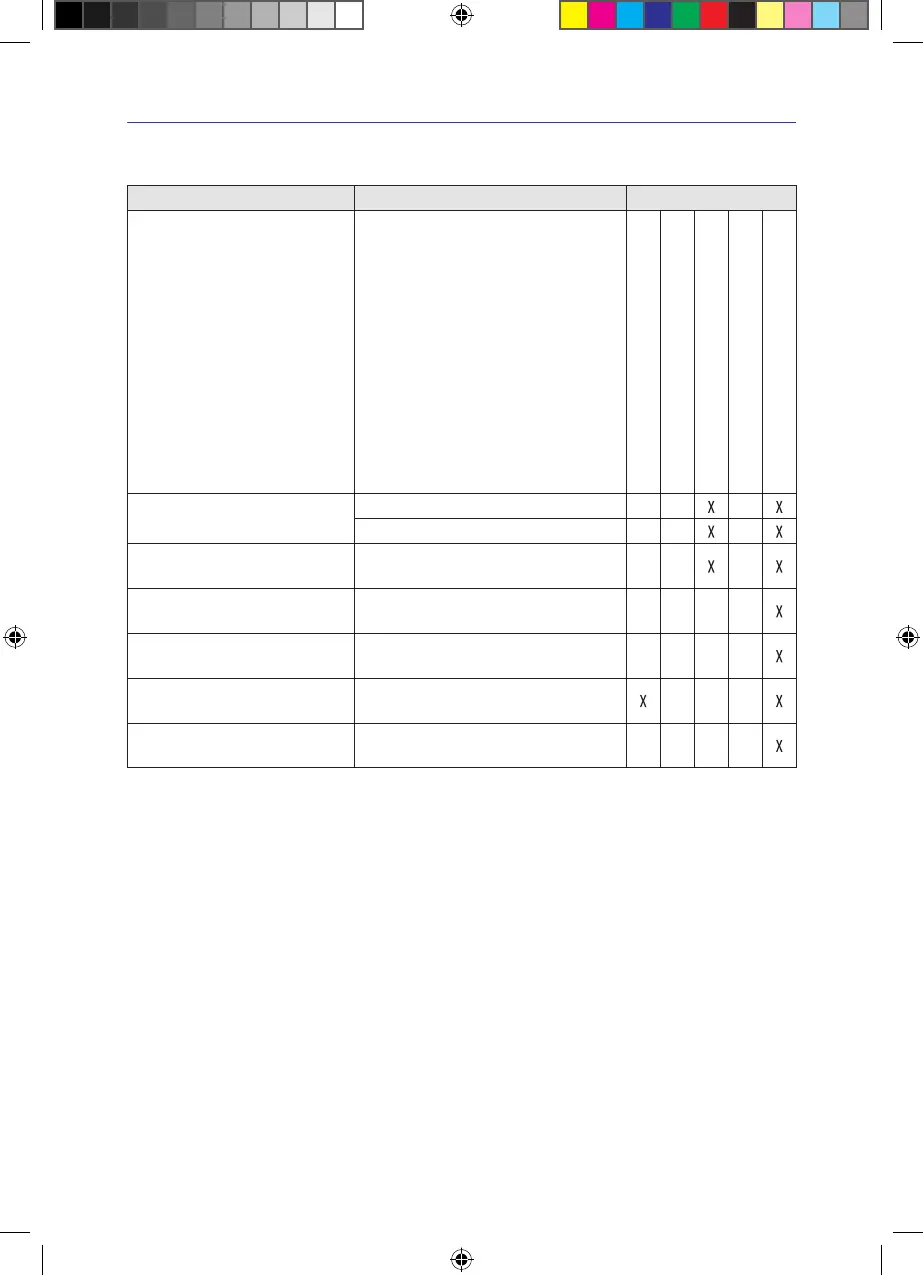
What Type When
After each dressing change
Daily
Weekly
Monthly
After each patient
VivanoTecPro Manual cleaning by wiping
Manual disinfection by wiping
VivanoTecExudateCanister Single-use product, not suitable for
reprocessing� Replace after use
VivanoTecShoulder Strap Single-use product, not suitable for
reprocessing� Replace after use
VivanoTec Bag Single-use product, not suitable for
reprocessing� Replace after use
VivanoMedFoamKit Single-use product, not suitable for
reprocessing� Replace after use
VivanoTecPro carry case
including inserts
Manual cleaning by wiping and disinfection
by wiping
11. Maintenance and service
11.1 Basic information
The negative pressure unit and its application components must be regularly and thoroughly cleaned� The negative
pressure unit must be operated only in accordance with the operating instructions� Comply with all national and
international regulations applicable to your institution�
11.2 Recurrent Tests and Repairs
The complete unit including power supply shall be sent to the manufacturer or authorised service partner for recurrent
tests every three years� Should repairs become necessary, please contact the manufacturer or an approved service partner�
Please contact the manufacturer or authorised service partner by telephone before sending the unit�
IFU_4095080_0302884_190824_EN.indd 36IFU_4095080_0302884_190824_EN.indd 36 19.08.24 12:3119.08.24 12:31

 Loading...
Loading...