B.14.1 Solvent miscibility
Before you change solvents, refer to the table below to determine solvent miscibility. Be aware of
these effects:
• Changes involving two miscible solvents can be made directly. Changes involving two
solvents that are not totally miscible (for example, from chloroform to water) require an
intermediate solvent like n-propanol.
• Temperature affects solvent miscibility. If you are running a high-temperature application,
consider the effect of the higher temperature on solvent solubility.
• Buffers dissolved in water can precipitate when mixed with organic solvents.
• When you switch from a strong buffer to an organic solvent, thoroughly flush the system with
distilled water before you add the organic solvent.
Note: λ cutoff is the wavelength at which the absorbance of the solvent equals 1 AU.
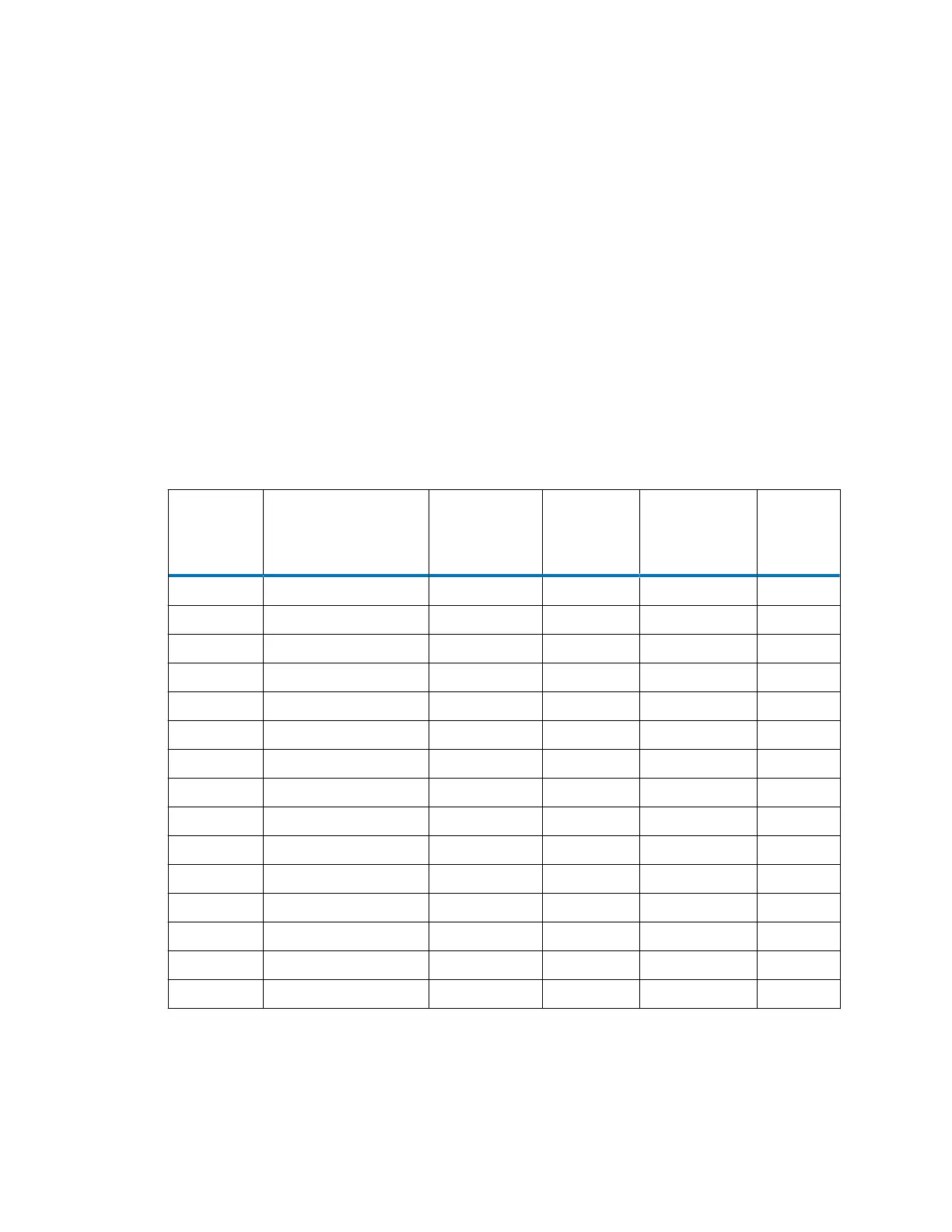
Table B–2: Solvent miscibility
Polarity
index
Solvent Viscosity cP,
20 °C (at 1
atm)
Boiling
point °C (at
1 atm)
Miscibility
number (M)
λ cutoff
(nm)
0.0 N-hexane 0.313 68.7 29 —
1.8 Triethylamine 0.38 89.5 26 —
4.2 Tetrahydrofuran (THF) 0.55 66.0 17 220
4.3 1-propanol 2.30 97.2 15 210
4.3 2-propanol 2.35 117.7 15 —
5.2 Ethanol 1.20 78.3 14 210
5.4 Acetone 0.32 56.3 15, 17 330
5.5 Benzyl alcohol 5.80 205.5 13 —
5.7 Methoxyethanol 1.72 124.6 13 —
6.2 Acetonitrile 0.37 81.6 11, 17 190
6.2 Acetic acid 1.26 117.9 14 —
6.4 Dimaethylformamide 0.90 153.0 12 —
6.5 Dimethlysulfoxide 2.24 189.0 9 —
6.6 Methanol 0.60 64.7 12 210
9.0 Water 1.00 100.0 — —
B.14.1.1 Using miscibility numbers (M-numbers)
Use miscibility numbers (M-numbers) to predict the miscibility of a liquid with a standard solvent.
August 8, 2016, 715005049 Rev. C
Page 94

 Loading...
Loading...









