A-37
Appendix A
Instructions for Use Manual Appendix A
the capillary test. Medical conditions and medications were
unavailable for critical care patients in this study.
Clinical Study #3 was a real world clinical review of 14,645
critical care patients throughout all critical care settings that
had a glucose test performed using a capillary whole blood
specimenobtainedbyngerstickandaplasmaglucose
test performed in the central laboratory within 15 minutes of
the capillary test. Medical conditions and medications were
unavailable for critical care patients in this study.
The capillary whole blood glucose results were compared
to plasma glucose results obtained from arterial or venous
specimens measured on an IDMS traceable Roche Cobas
Modular P800 hexokinase method (Roche Diagnostics,
Indianapolis, IN).
A.8.3 Clinical Study Performance
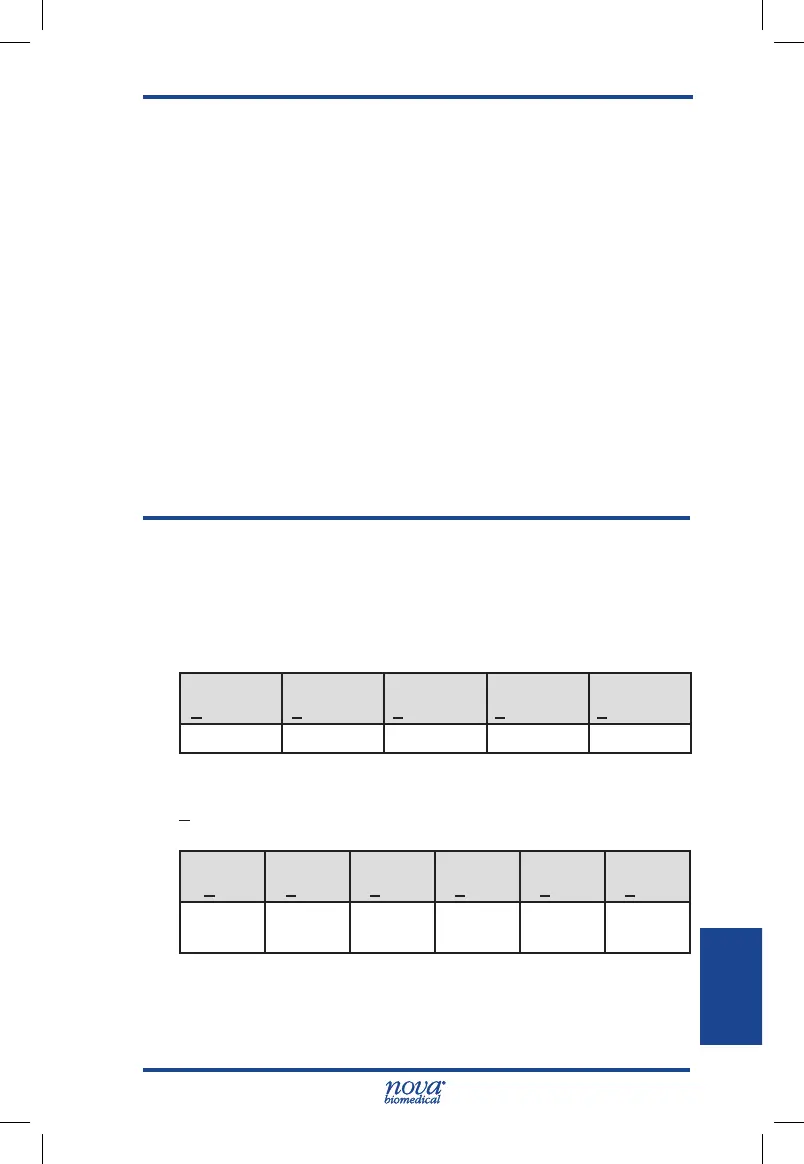
Table A-55 Study #1 (Prospective Study)
Fingertip capillary samples with glucose concentrations
<75 mg/dL
Within
+5 mg/dL
Within
+10 mg/dL
Within
+12 mg/dL
Within
+15 mg/dL
Exceeds
+15 mg/dL
1/1(100%) 1/1(100%) 1/1(100%) 1/1(100%) 0/1(0%)
Fingertip capillary samples with glucose concentrations
>75 mg/dL
Within
+5%
Within
+10%
Within
+12%
Within
+15%
Within
+20%
Exceeds
+20%
277/567
(48.9%)
450/567
(79.4%)
484/567
(85.4%)
516/567
(91.0%)
549/567
(96.8%)
18/567
(3.2%)
StatStrip Glu IFU 1.86 PN55848E EN 510K.indd 37 9/13/18 8:29 AM
 Loading...
Loading...