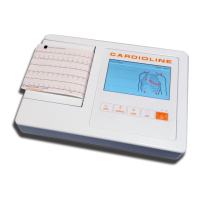
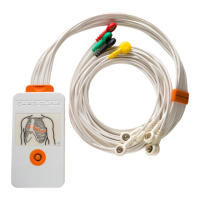
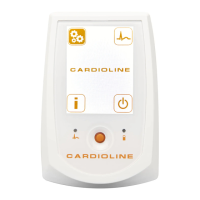
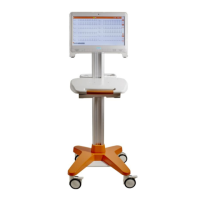
ECG100+ - ECG200+ - ECG100S - ECG200S
13. TECHNICAL SPECIFICATION
“A 0,3 mV × s (3 mV for 100 ms) impulse input shall not produce a displacement greater than 0.1 mV
outside the region of the impulse.”
The acquisition (sampling and filtering) system of the device complies with AHA recommendations (Paul
Kligfield et al., Circulation 2007) for both paediatric and adult ECGs. The high frequency response of the
system is 150Hz or 300Hz, depending on the filter applied.
The device also offers the option of applying, only in display and printing mode, digital linear-phase filters
with a cutoff frequency of 25Hz or 40Hz, which reduce the bandwidth of the printed signal in order to
reduce the effects of high frequency noise ("muscular" noise). As a consequence of the application of these
filters, the resulting signal no longer meets the minimum requirements on the high frequency response
reported in the above recommendations.
The device can also be configured to selectively eliminate interference induced by the electricity network
(50Hz or 60Hz, depending on the country where the device is installed).
The network filter complies with the requirements of IEC 60601-2-25 2nd Ed.
13.2. Harmonised standards applied
Medical devices - Symbols to be used with medical device
labels, labelling and information to be supplied - Part 1:
General requirements
Information supplied by the manufacturer of medical devices
Medical devices - Quality management systems -
Requirements for regulatory purposes
Medical devices - Application of risk management to medical
devices)
Medical electrical equipment - Part 1: General requirements
for basic safety and essential performance
Medical electrical equipment - Part 1: General requirements
for basic safety and essential performance - Collateral
standard: Electromagnetic compatibility - Requirements and
tests
Medical electrical equipment - Part 1: General safety
requirements - Collateral standard: Usability
Medical electrical equipment - Part 2-25: Particular
requirements for the safety of electrocardiographs.
Medical device software - Software life cycle processes EN
62304:2006/AC:2008
 Loading...
Loading...