This document contains proprietary and confidential information of Integra LifeSciences Corporation. Integra’s confidential information may not be used, disclosed or reproduced without the prior wrien consent of Integra LifeSciences Corporation.
CUSA® Clarity System Service and Maintenance Manual Page 8 of 117 0645403-2-EN
Chapter 1
1.1 Intended Audience
This manual provides servicing information for the CUSA® Clarity Surgical Ultrasonic Aspirator system. The manual is intended for use by a certified
biomedical technician, as well as Integra authorized service partner responsible for the safe handling and maintenance of the CUSA® Clarity console.
1.2 Intended Usage of this Manual
This Service and Maintenance Manual describes how to perform basic cleaning, inspection, maintenance, troubleshooting, and non-invasive repair of
the CUSA® Clarity console. This manual provides specific reference materials that may be used to train new technicians in performance of functional
tests, and field replacement item installation. The manual also provides functional testing procedures for operational verification of the console prior to
clinical use.
1.3 Trademark Acknowledgments
Integra, the Integra logo, CUSA, and Tissue Select are registered trademarks of Integra LifeSciences Corporation or its subsidiaries in the United States
and/or other countries. ShearTip, MicroTip, and CUSA Quick Connect are trademarks of Integra LifeSciences Corporation or its subsidiaries.
Sani-Cloth is a trademark of Professional Disposables International, Inc. neodisher is a trademark of Chemische Fabrik Dr. Weigert GmbH & Co. KG. All
other trademarks and trade names are the property of their respective owners.
CAUTION
Federal (USA) law restricts this device to sale by or on the order of a physician.
1.4 Manufacturer
Integra LifeSciences (Ireland) Limited IDA Business & Technology Park Sragh Tullamore, County Offaly, Ireland Integra LifeSciences Corporation
Plainsboro, NJ 08536 USA; 1-800-997-4868
© 2017 Integra LifeSciences Corporation. All Rights Reserved. Do not reproduce the contents of this publication in any form without the wrien
permission of Integra.
1.5 Patent Information
U.S. Patents 6,499,358; 8,118,823; 8,518,066; 9,421,027; additional patent(s) pending.
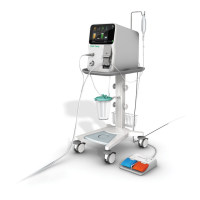
1.6 System Features
The CUSA Clarity system includes several important components and features:
• Console with touchscreen interface
• Cart
• 36 kHz handpiece
• A variety of surgical tips that aach to the handpiece:
o Sterile, single-use tips
o Non-sterile, extended use tips (EUT)
• Tissue Select® feature, which increases the selectivity of the surgical tip, allowing greater control and precision
 Loading...
Loading...