52
Maintenance
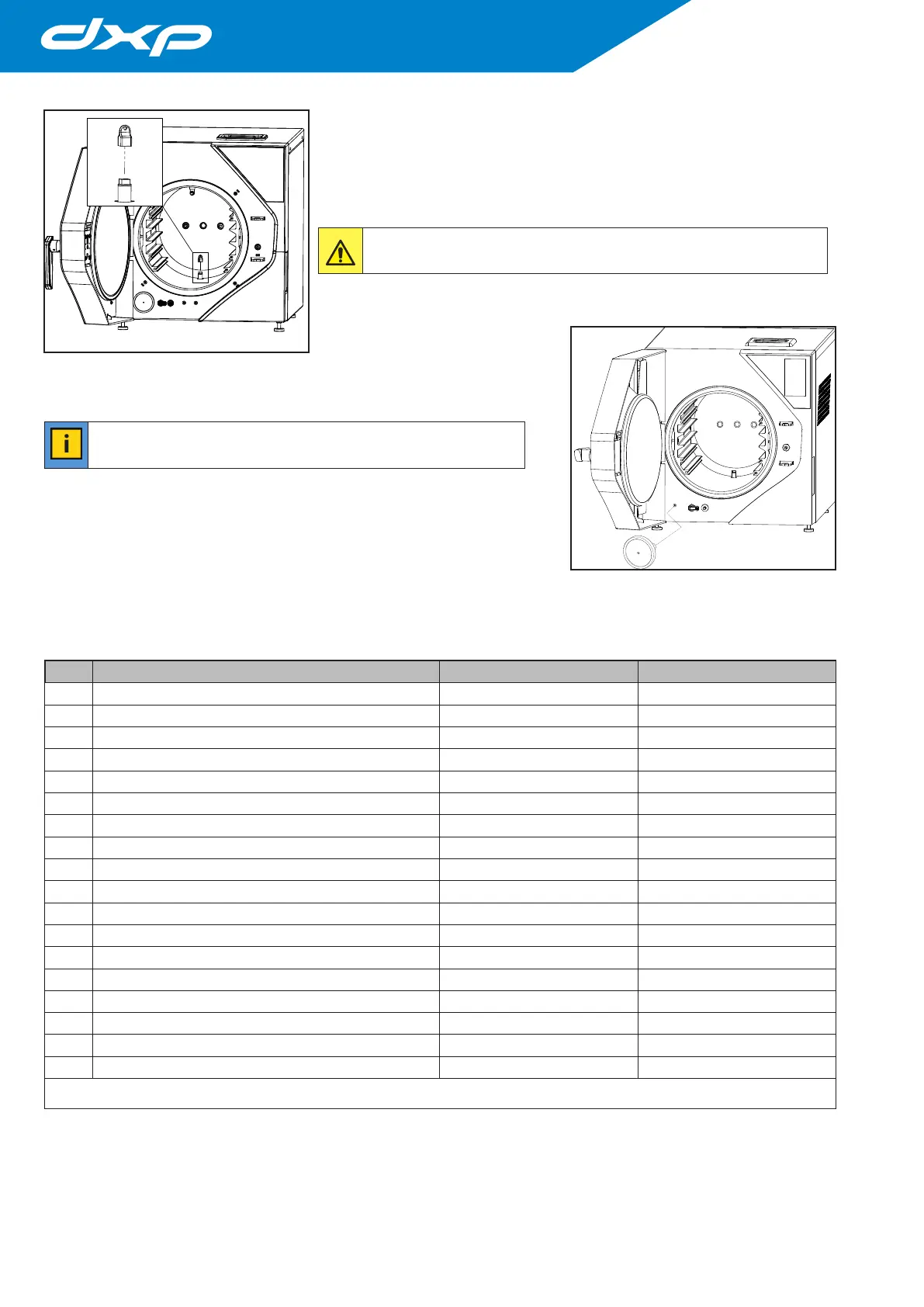
7.1.2 H2O chamber filter clean and replacement
To clean or replace the H20 lter in the chamber, it must be opened
the hatch (unlocking it with the appropriate command on the screen) e
unscrewing the lter at the bottom using the key supplied in the autoclave
accessories kit.
Pay attention: possible hot surfaces.
7.1.3 Replacing the bacteriological filter
Replace the bacteriological filter with the device switched off.
Unscrew the bacteriological filter and replace it with a new one. Install the
filter in its housing, taking care to ensure that it is fully screwed in.
7.2 Scheduled maintenance
The following table indicates the scheduled maintenance operations to be carried out on the device by qualified
and authorized personnel at the customer’s premises, with the relative deadlines.
Pos Check/activity Responsible Frequence
1 Door adjustment 1 Year / 1.000 cycles
- Gasket replacement Maintainer / Technician 1 Year / 1.000 cycles
- Disk-door clearance check Maintainer / Technician 1 Year / 1.000 cycles
- Lubrification Maintainer / Technician 1 Year / 1.000 cycles
- Component wear check Maintainer / Technician 1 Year / 1.000 cycles
- Closing pin replacement (if necessary) Technician 2 Years / 2.000 cycles
2 Calibration Maintainer / Technician 1 Year
3 Filter cleaning / replacement 1 Year / 1.000 cycles
- Chamber cylindrical filter replacement Operator 1 Year / 1.000 cycles
- Clean water tank filter replacement Operator 1 Year / 1.000 cycles
4 Pump feature check Technician 1 Year / 1.000 cycles
5 Tanks cleaning Maintainer / Technician 1 Year / 1.000 cycles
6 Radiator cleaning Maintainer / Technician 1 Year / 1.000 cycles
7 Non-return valve replacement Technician 2 Years / 2.000 cycles
8 Micropump replacement Technician 2 Years / 2.000 cycles
9 Electric closing piston check / lubricate Maintainer / Technician 2 Years / 2.000 cycles
10 Radiator outlet brass filter inspection / cleaning Maintainer / Technician 2 Years / 2.000 cycles
11 Vacuum Pump Maintenance Technician 2 Years / 2.000 cycles
NOTE: refer to the legislation and instructions for each individual country.
7.3 Periodic validation / requalification of the sterilization process
Th e sterilizer pu rchased c omplie s with the ap plicab le requi rements of th e safet y standa rds in force, a nd the para meters
set by the manufacturer are designed to ensure the sterility of the load in the conditions indicated in the manual.
To ensure process safety over time, it is necessary to check, periodically (1 year), the process parameters (pressure and
temperature), making sure that they remain within the limits set by the European reference standards EN 17665. and

 Loading...
Loading...