20 Chapter 1
P/N 130859-US, Manual Revision: AA Haemonetics
®
Cell Saver
®
Elite
®
+ User Manual
Device Specifications
Note: The use of materials not provided or recommended by Haemonetics is
the sole responsibility of the end-user, and the end-user will be responsible for
any personal injury and/or property damage related to such use.
Device
Classification
The Cell Saver Elite+ device is classified as a continuous operation, Class I,
Type CF, IPX1 device, in accordance with IEC/EN 60529 and 60601 standards
for medical electrical equipment.
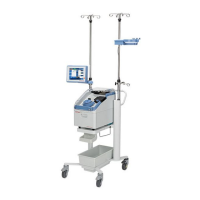
Physical
Specifications
The approximate dimensions and weight of the Cell Saver Elite+ device are as
follows:.
The noise level of the Cell Saver Elite+ device is < 70 dB.
Environmental
Specifications
The following environmental conditions should be respected pertaining to
operation and storage of the Cell Saver Elite+ device.
Alert: Equipment not suitable for use in the presence of a flammable
anesthetic mixture with air or with oxygen or nitrous oxide.
Note: Store disposable components in a dry place away from solvent vapors
and extremes of temperature.
Table 8, Physical Specifications
Depth/in. (cm) Height/in. (cm) Width/in. (cm)
Device Alone 21.5 in.
(54.6 cm)
16.5 in.
(41.9 cm)
11.75 in.
(29.8 cm
Device With Cart
IV poles extended 26.5 in.
(67.3 cm)
72 in. (182.9 cm) 21 in.
(53.3 cm)
IV poles down 26.5 in.
(67.3 cm)
48 in. (121.9 cm) 21 in.
(53.3 cm)
Weight of device 56 lbs. (25 kg)
Weight of cart 39 lbs. (18 kg)
Safe working load
of system
157 lbs. (71 kg)